Watch 02: April 2026
The HIV/HCV Co-Infection Watch is a project of the Community Access National Network (CANN) designed to research, monitor, and report on HIV and Hepatitis C (HCV) co-infection in the United States. The April 2026 Watch includes timely updates herein. To read the project disclaimer and/or methodology, CLICK HERE.
1. FINDINGS
The following is a summary of the key findings for April 2026:
AIDS Drug Assistance Programs:
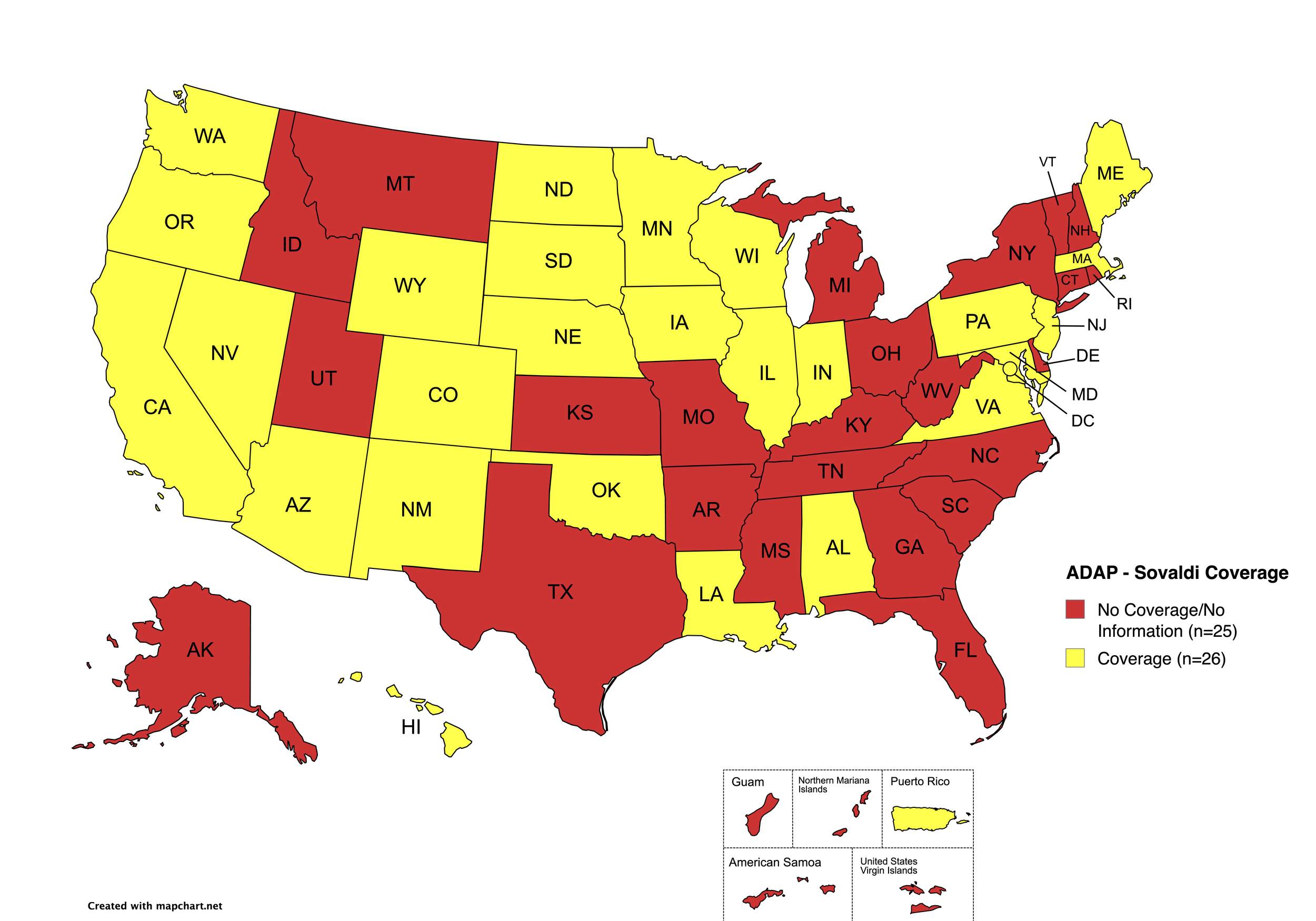
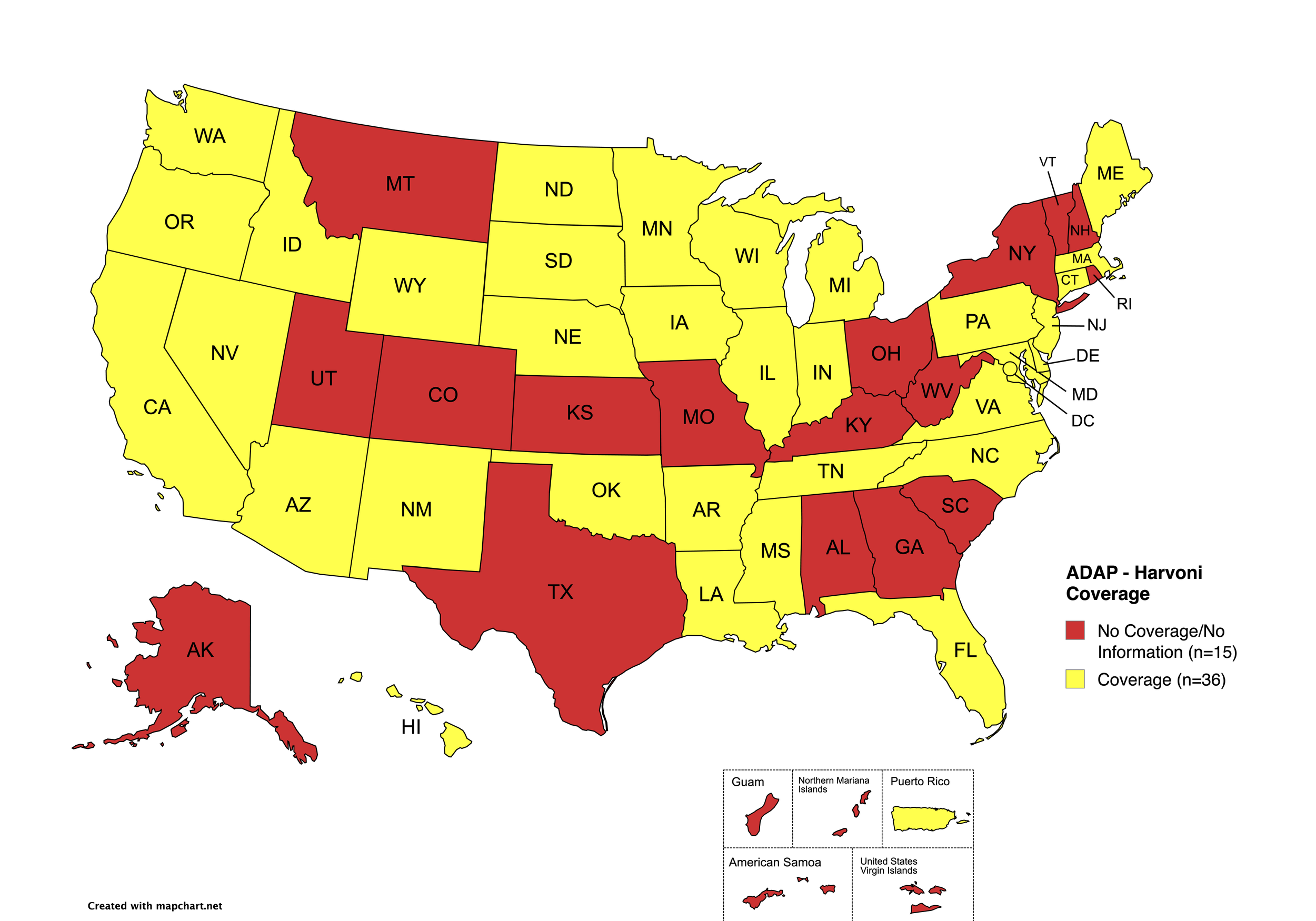
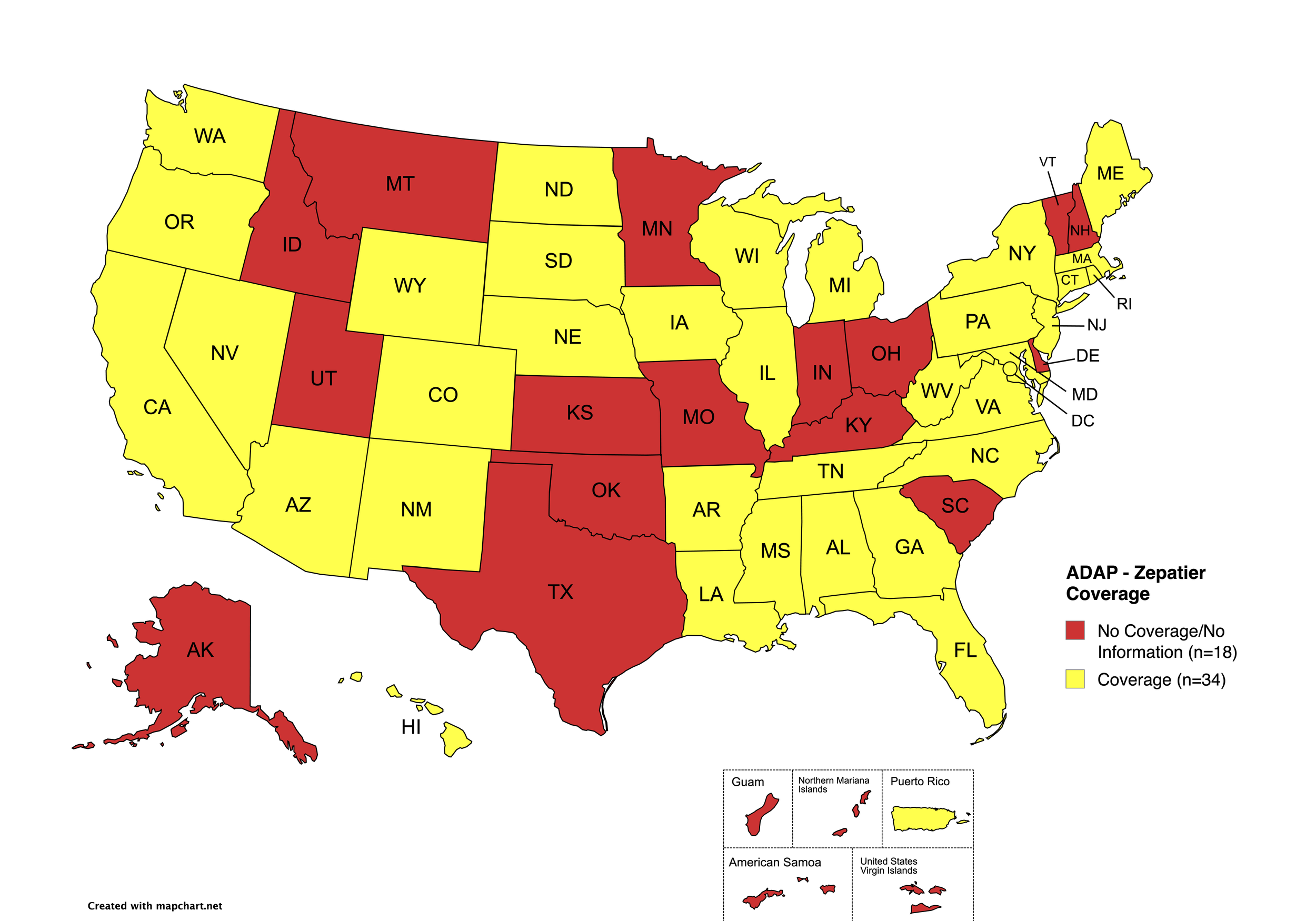
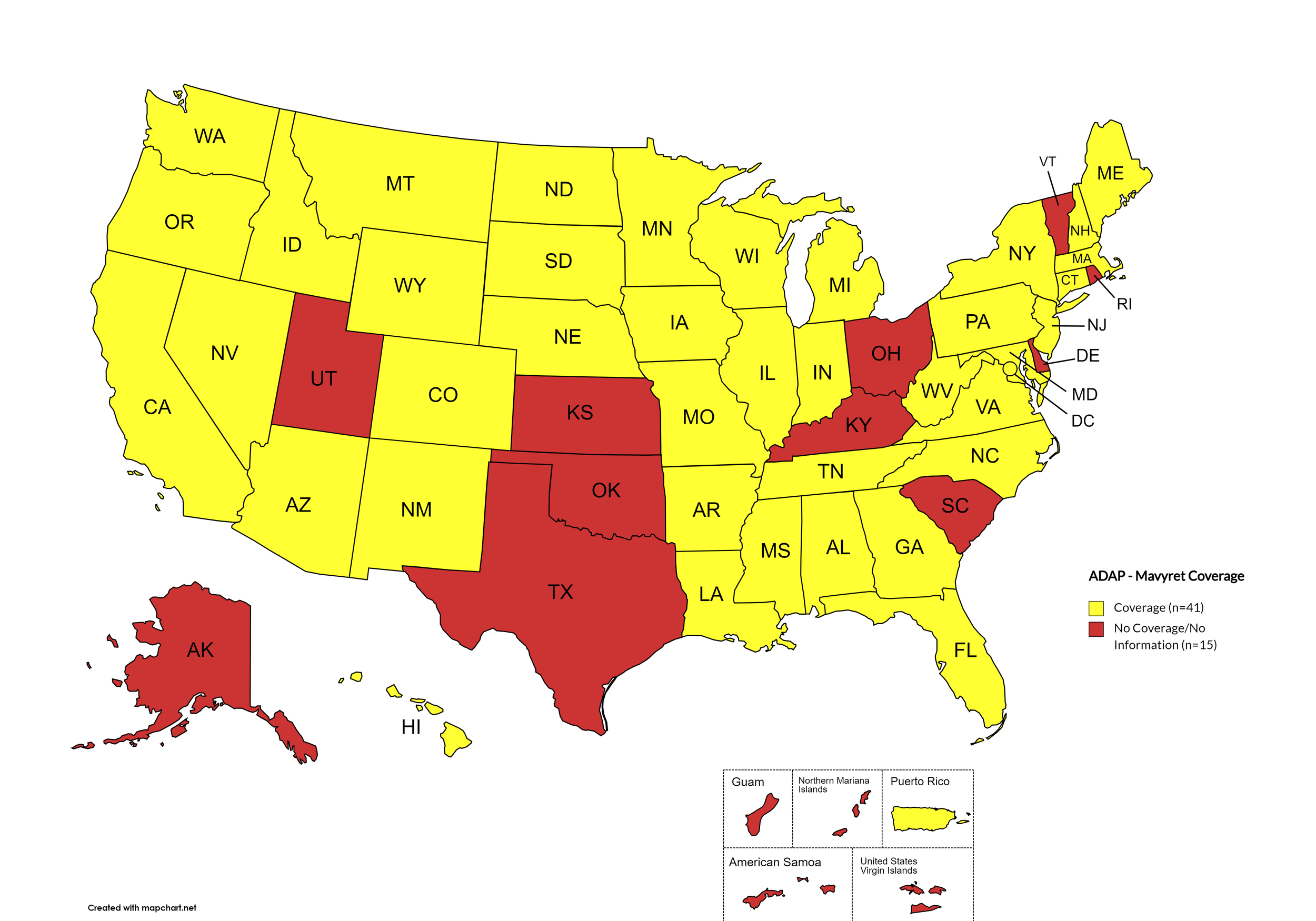
There are 56 state and territorial AIDS Drug Assistance Programs (ADAPs) in the United States, 48 of which offer some form of coverage for Hepatitis C (HCV) treatment. Of those programs, 45 have expanded their HCV coverage to include the Direct-Acting Antiviral (DAA) regimens that serve as the current Standard of Care (SOC) for Hepatitis C treatment. Two (2) programs offer only Basic Coverage, and eight (8) programs offer No Coverage. Three (3) programs cover only select Direct-Acting Antivirals with significant restrictions. Three (3) territories – American Samoa, Marshall Islands, and Northern Mariana Islands – are not accounted for in this data. The most significant federal development was the introduction of the Cure Hepatitis C Act of 2025 in June, proposing a subscription model for HCV medications that could transform access nationwide. A state-by-state Drug Formulary breakdown of coverage is included in the April 2026 Updates, with accompanying drug-specific maps in Figures 1 – 10.
Medicaid Programs:
There are 59 state and territorial Medicaid programs in the United States, and data is represented for all 50 states and the District of Columbia. As of October 2025, all 50 states and the District of Columbia continue to offer Expanded Coverage for hepatitis C treatment. The coverage landscape has seen significant changes since the previous report: Louisiana removed prior authorization requirements for generic HCV medications; Ohio Medicaid began offering HCV treatment for the first time with ribavirin, Mavyret, Pegasys, and generic Epclusa; Nevada substantially reduced its formulary by removing Sovaldi, Harvoni, Zepatier, Vosevi, and generic Harvoni; and South Carolina added Epclusa to its preferred list. Importantly, all state Medicaid programs have maintained their removal of fibrosis restrictions for initial treatment, and no states currently require sobriety as a prerequisite for hepatitis C treatment. Rhode Island remains the only state that does not cover generic Epclusa. A complete state-by-state PDL breakdown of coverage is included in the April 2026 Updates, with accompanying drug-specific maps in Figures 11 – 20.
Patient Assistance Programs:
Our April 2026 analysis of Patient Assistance Programs (PAPs) for hepatitis C treatments reveals mixed developments in program availability. The HealthWell Foundation had reopened the Hepatitis C fund with a maximum award of $10,000, providing crucial financial support, but it is still closed. However, significant changes occurred with Gilead's Support Path program, which implemented major modifications on May 5, 2025, transitioning from retail pharmacy to mail order delivery and discontinuing free medications for several products due to generic availability. The Patient Advocate Foundation's Co-Pay Relief program has reopened to new HCV applications, while The Assistance Fund remains in waitlist status. The PAN Foundation adjusted grant amounts beginning January 1, 2025, in response to Medicare Part D's new $2,000 out-of-pocket cap. However, it is currently closed to new or renewing members. It is only accepting eligible waitlist recipients. These program changes reflect the evolving landscape of patient assistance as generic medications become more widely available and federal healthcare policies shift.
Harm Reduction Programs:
Syringe Services Programs: Forty-three (43) states, the District of Columbia and two (2) territories currently have Syringe Services Programs (SSPs) in place, regardless of the legality. Seven (7) states without SSPs are Alabama, Idaho, Kansas, Mississippi, Nebraska, South Dakota, and Wyoming. Idaho's situation reflects the most significant change, having repealed its Syringe and Needle Act (House Bill 617) effective July 1, 2024, becoming the first state to completely eliminate authorized syringe services programs.
Expanded Naloxone Access: All fifty (50) states and the District of Columbia have health department distribution programs for Naloxone and/or allow Medicaid coverage of Naloxone. No states have restricted naloxone access, with innovative distribution methods emerging including vending machine programs in Denver (2,100+ boxes distributed), St. Louis County (44 new machines), and California's direct-to-consumer CalRx program offering naloxone at $24 per twin-pack.
Safe Consumption Sites: Two (2) states explicitly authorize or have authorized pilot projects for Safe Consumption Sites (SCSs): New York (operational since November 2021) and Rhode Island (opened December 2024). Rhode Island's first state-sanctioned overdose prevention center reported over 500 visitors and 27 overdose deaths prevented in its first six months of operation. The legislature approved a two-year extension of the pilot program in April 2025.
Updated Paraphernalia Laws: Forty-nine (49) states have modernized their criminal codes to allow for possession of testing strips and may also have health department programs distributing testing strips. Indiana decriminalized fentanyl test strips effective July 1, 2025, through House Bill 1167. Iowa remains the only state where testing strips are still explicitly categorized as illegal paraphernalia.
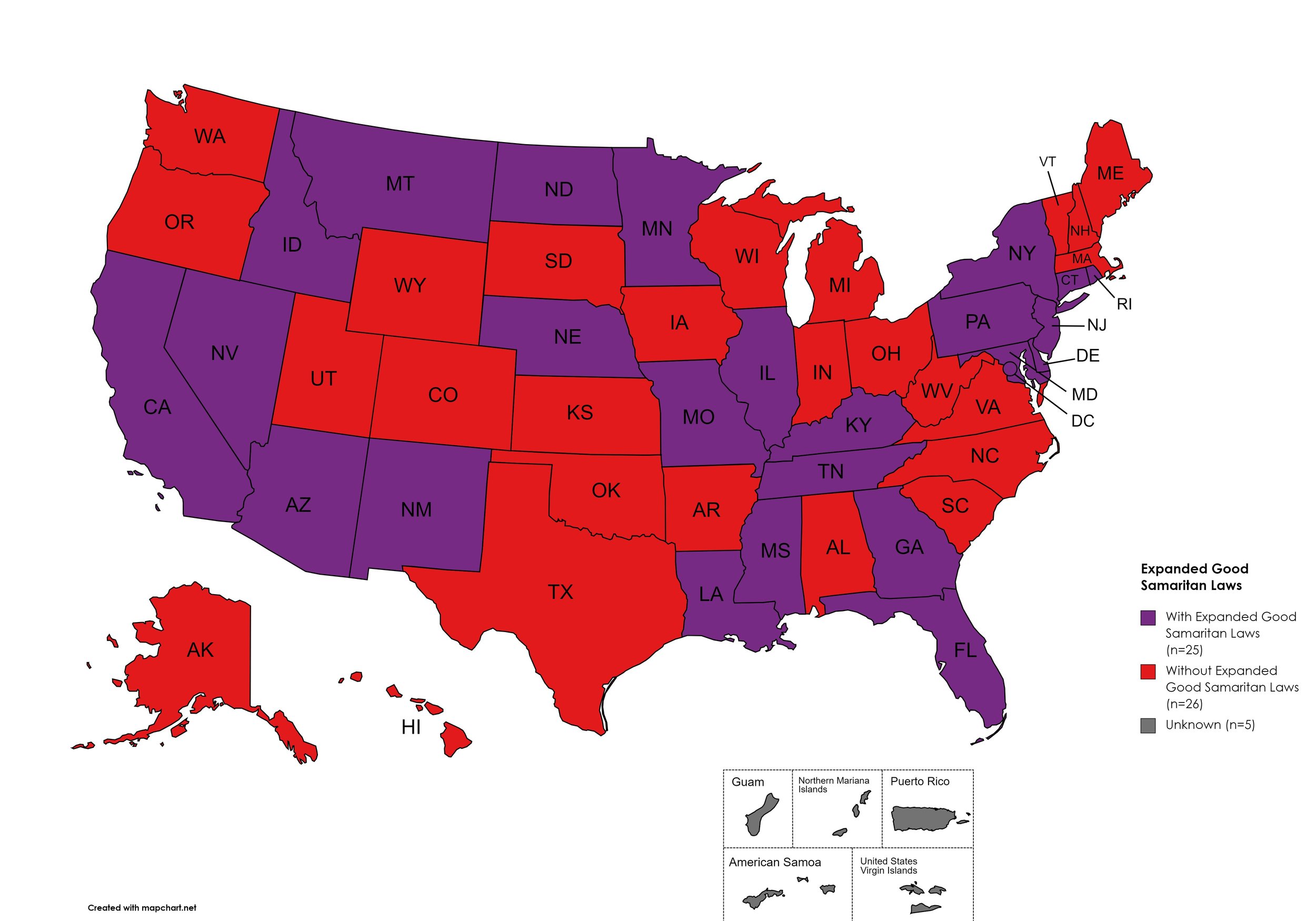
Good Samaritan Laws: Twenty-five (25) states have Good Samaritan laws or statutes that provide some level of protection for those seeking or giving assistance during a drug overdose, regardless of possession of controlled substances or consumption of illegal or illicit substances. No new states added Good Samaritan protections during the research period.
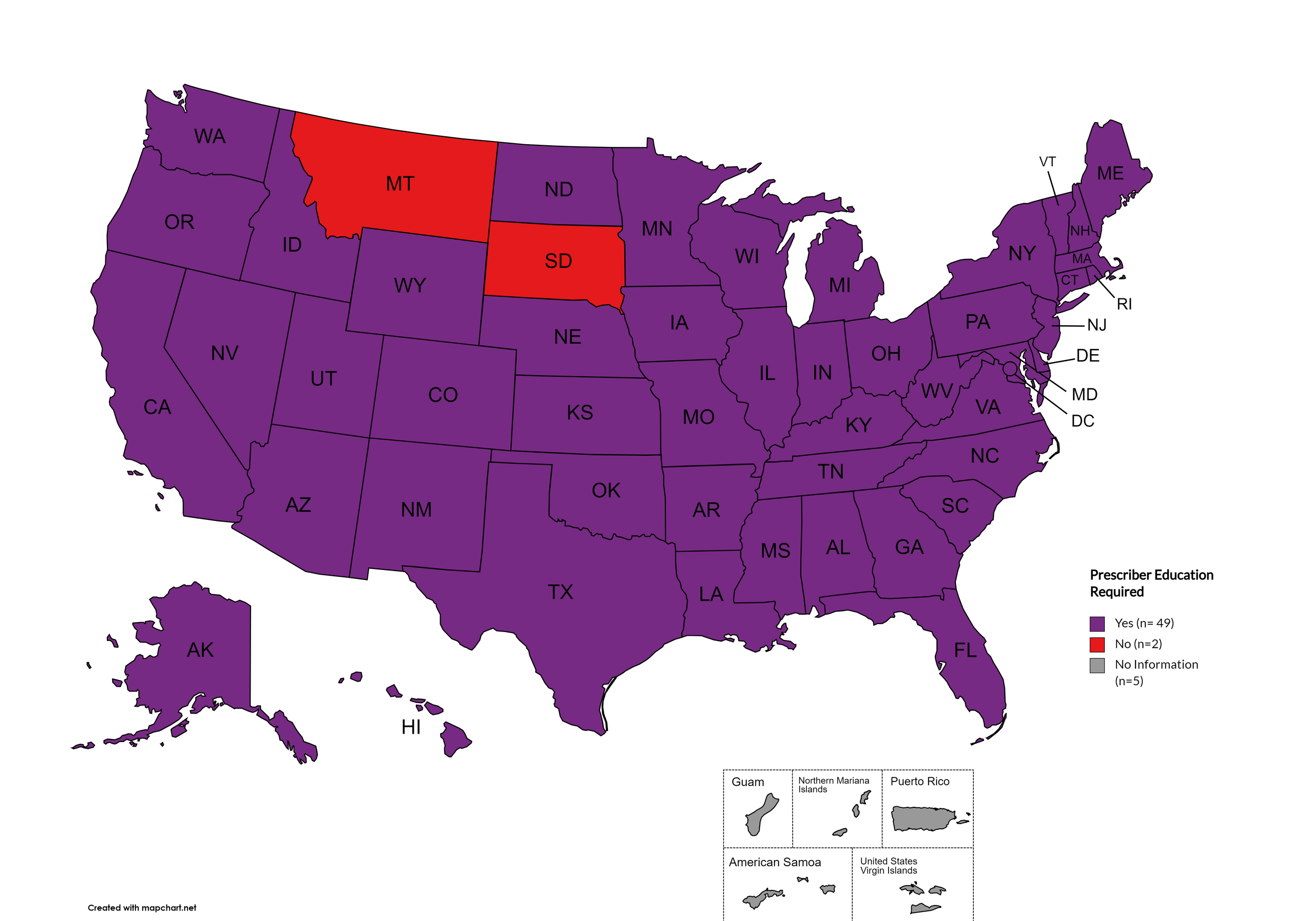
Prescriber Education: Forty-eight (48) states and the District of Columbia require, through legislative action or regulatory or licensing bodies, that prescribing physicians attend mandatory and continuing opioid prescribing or harm reduction education sessions. Montana and South Dakota remain the only states without these requirements. A state-by-state program breakdown is included in the October 2025 Updates, with accompanying maps in Figures 21-26.
2. AIDS DRUG ASSISTANCE PROGRAMS (ADAPs) & HCV THERAPIES
Of the 56 respective state and territorial ADAPs, only 8 (KS, KY, OH, UT, VT, GU, PW, VI) do not offer any coverage for HCV drug therapies. States whose formularies are not available on the state-run website have been checked against the most recent National Alliance of State and Territorial AIDS Directors (NASTAD) formulary database (last updated January 1, 2025). The data presented are current as of April 28, 2026.
April 2026 Updates:
Basic Coverage
States with Basic HCV Medications Coverage: CA, AL, AK, AZ, AR, CO, CT, DE, FL, GA, HI, IL, IN, IA, LA, ME, MD, MA, MI, MN, MO, MT, NE, NV, NH, NJ, NM, NY, NC, ND, OK, OR, PA, RI, SC, SD, TN, VA, WA, WV, WY, D.C.
States without Basic HCV Medications Coverage: ID, KS, KY, MS, OH, TX, UT, VT, WI
Territories with Basic HCV Medications Coverage: P.R.
Figure 1. April 2026 ADAP Coverage - Basic
Map Key: Yellow = Basic Coverage; Red = No Basic Coverage/No Information regarding Basic Coverage
Sovaldi
States with Sovaldi Coverage: AZ, CA, CO, HI, IL, IN, IA, LA, ME, MD, MA, MN, NE, NV, NJ, NM, ND, OK, OR, PA, SD, VA, WA, WI, WY, D.C.
States without Sovaldi Coverage: AL, AK, AR, CT, DE, FL, GA, ID, KS, KY, MI, MS, MO, MT, NH, NY, NC, OH, RI, SC, TN, TX, UT, VT, WV
Territories with Sovaldi Coverage: P.R.
Figure 2. April 2026 ADAP Coverage - Sovaldi
Map Key: Yellow = Sovaldi Coverage; Red = No Sovaldi Coverage/No Information regarding Sovaldi Coverage
Harvoni
States with Harvoni Coverage: AZ, AR, CA, CO, CT, DE, FL, HI, ID, IL, IN, IA, LA, ME, MD, MA, MI, MN, MS, NE, NV, NJ, NM, NC, ND, OK, OR, PA, SD, TN, VA, WA, WI, WY, D.C.
States without Harvoni Coverage: AL, AK, GA, KS, KY, MO, MT, NH, NY, OH, RI, SC, TX, UT, VT, WV
Territories with Harvoni Coverage: P.R.
Figure 3. April 2026 ADAP Coverage - Harvoni
Map Key: Yellow = Harvoni Coverage; Red = No Harvoni Coverage/No Information regarding Harvoni Coverage
Zepatier
States with Zepatier Coverage: AL, AZ, AR, CA, CO, FL, GA, HI, IL, IA, LA, ME, MD, MA, MI, MS, NE, NV, NJ, NM, NY, NC, ND, OR, PA, SD, VA, WA, WV, WI, WY, D.C.
States without Zepatier Coverage: AK, CT, DE, ID, IN, KS, KY, MN, MO, MT, NH, OH, OK, RI, SC, TN, TX, UT, VT
Territories with Zepatier Coverage: P.R.
Figure 4. April 2026 ADAP Coverage - Zepatier
Map Key: Yellow = Zepatier Coverage; Red = No Zepatier Coverage/No Information regarding Zepatier Coverage
Epclusa
States with Epclusa Coverage: AZ, AR, CA, CO, CT, FL, HI, ID, IL, IN, IA, LA, ME, MD, MA, MI, MN, MS, MO, NE, NV, NJ, NM, ND, OR, PA, SD, TN, VA, WA, WI, WY
States without Epclusa Coverage: AL, AK, DE, GA, KS, KY, MT, NH, NY, NC, OH, OK, RI, SC, TX, UT, VT, WV, D.C.
Territories with Epclusa Coverage: P.R.
Figure 5. April 2026 ADAP Coverage - Epclusa
Map Key: Yellow = Epclusa Coverage; Red = No Epclusa Coverage/No Information regarding Epclusa Coverage
Vosevi
States with Vosevi Coverage: CA, CO, CT, FL, HI, ID, IL, IN, IA, LA, ME, MD, MA, NE, NV, NJ, NM, ND, OR, SD, TN, WA, WY
States without Vosevi Coverage: AL, AK, AZ, AR, DE, GA, KS, KY, MI, MN, MS, MO, MT, NH, NY, NC, OH, OK, PA, RI, SC, TX, UT, VT, VA, WV, WI, D.C.
Territories with Vosevi Coverage: P.R.
Figure 6. April 2026 ADAP Coverage - Vosevi
Map Key: Yellow = Vosevi Coverage; Red = No Vosevi Coverage/No Information regarding Vosevi Coverage
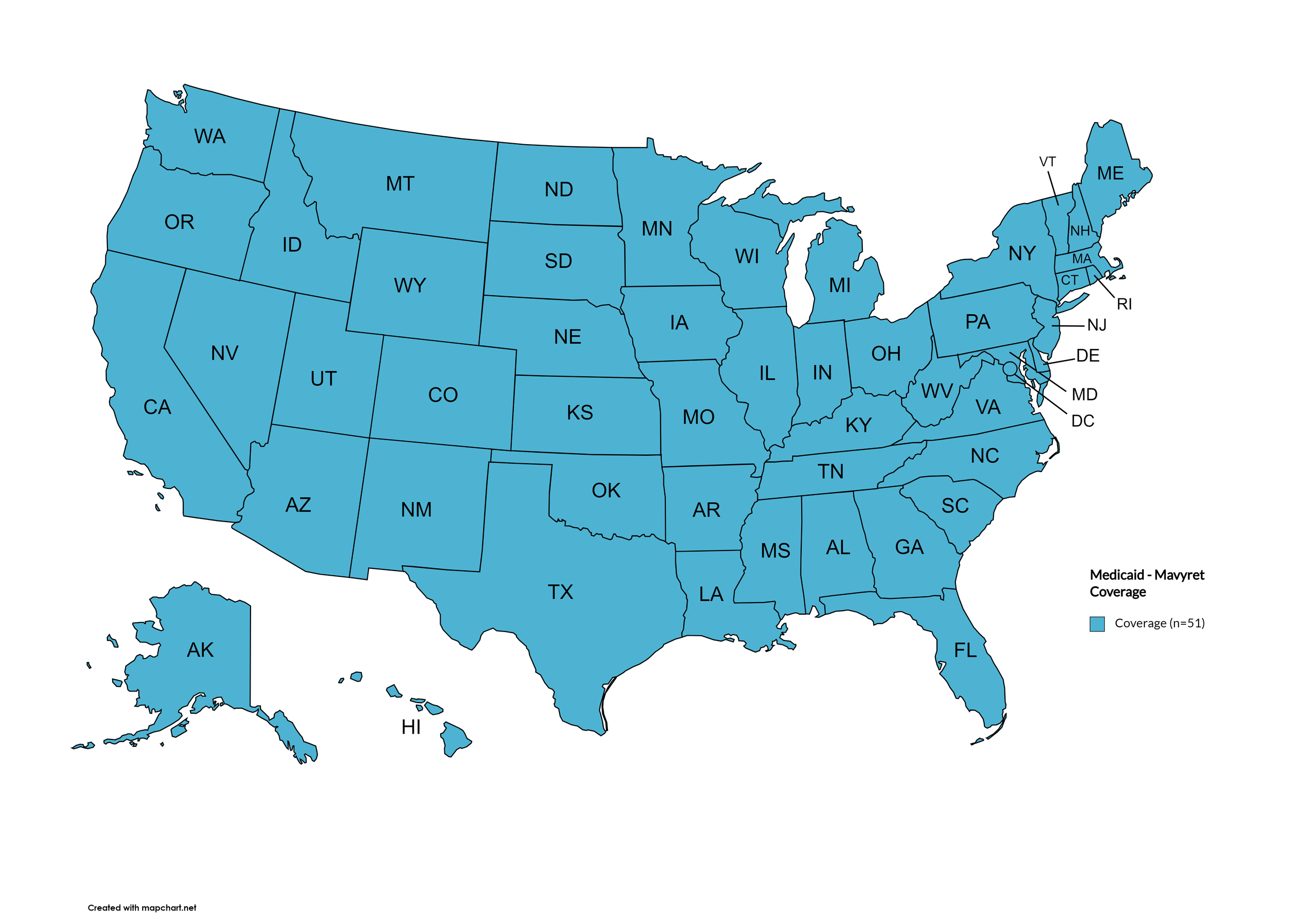
Mavyret
States with Mavyret Coverage: AL, AZ, AR, CA, CO, CT, FL, GA, HI, ID, IL, IN, IA, LA, ME, MD, MA, MI, MN, MS, MO, MT, NE, NV, NJ, NM, NY, NC, ND, OR, PA, SD, TN, VA, WA, WV, WI, WY, D.C.
States without Mavyret Coverage: AK, DE, KS, KY, NH, OH, OK, RI, SC, TX, UT, VT
Territories with Mavyret Coverage: P.R.
Figure 7. April 2026 ADAP Coverage - Mavyret
Map Key: Yellow = Mavyret Coverage; Red = No Mavyret Coverage/No Information regarding Mavyret Coverage
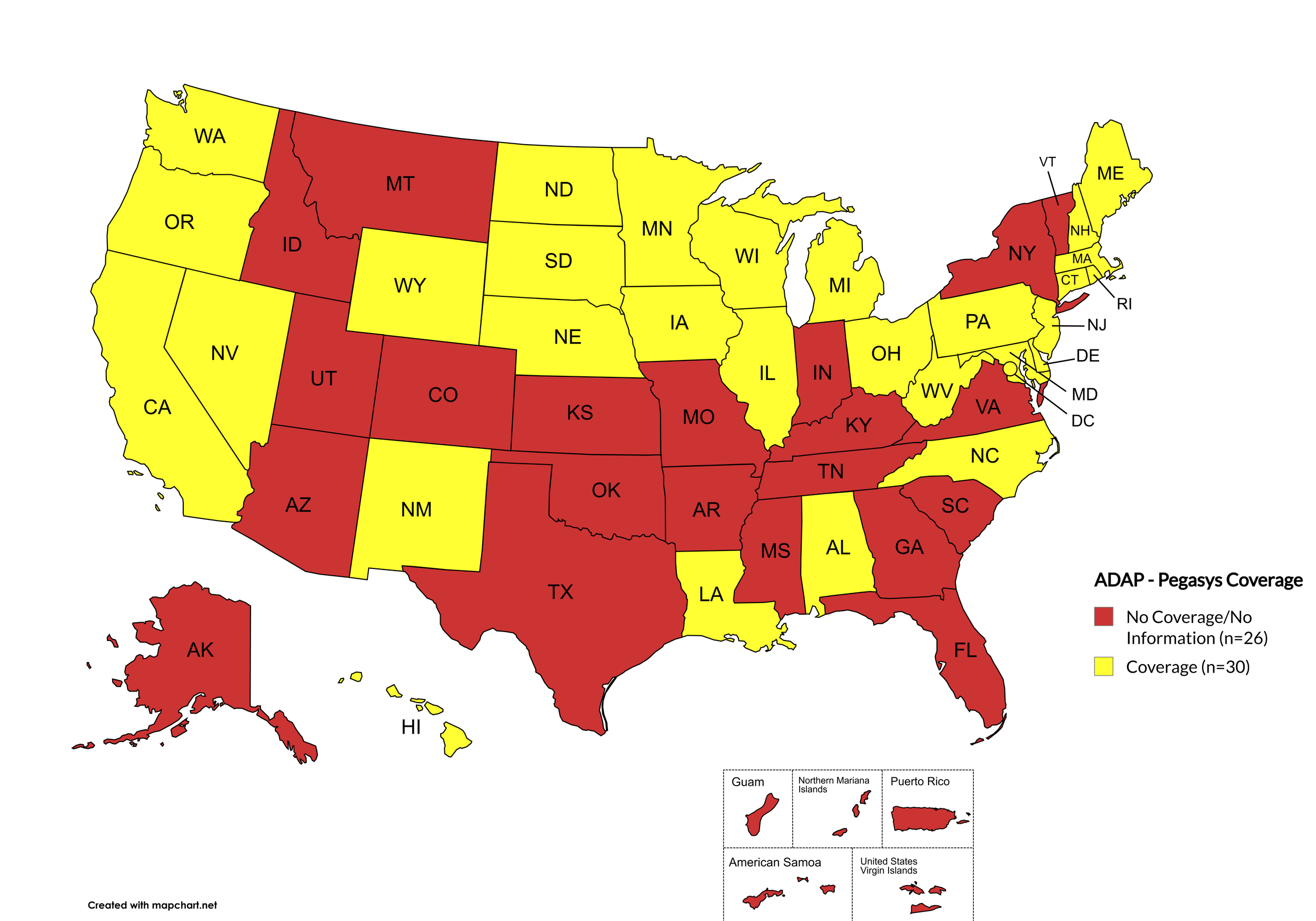
Pegasys
States with Pegasys Coverage: AL, CA, CT, DE, HI, IL, IA, LA, ME, MD, MA, MI, MN, NE, NV, NJ, NM, NC, ND, OH, OR, PA, RI, SD, WA, WV, WI, WY, D.C.
States without Pegasys Coverage: AK, AZ, AR, CO, FL, GA, ID, IN, KS, KY, MS, MO, MT, NH, NY, OH, OK, SC, TN, TX, UT, VT, VA
Territories with Pegasys Coverage: None/Unknown
Figure 8. April 2026 ADAP Coverage - Pegasys
Map Key: Yellow = Pegasys Coverage; Red = No Pegasys Coverage/No Information regarding Pegasys Coverage
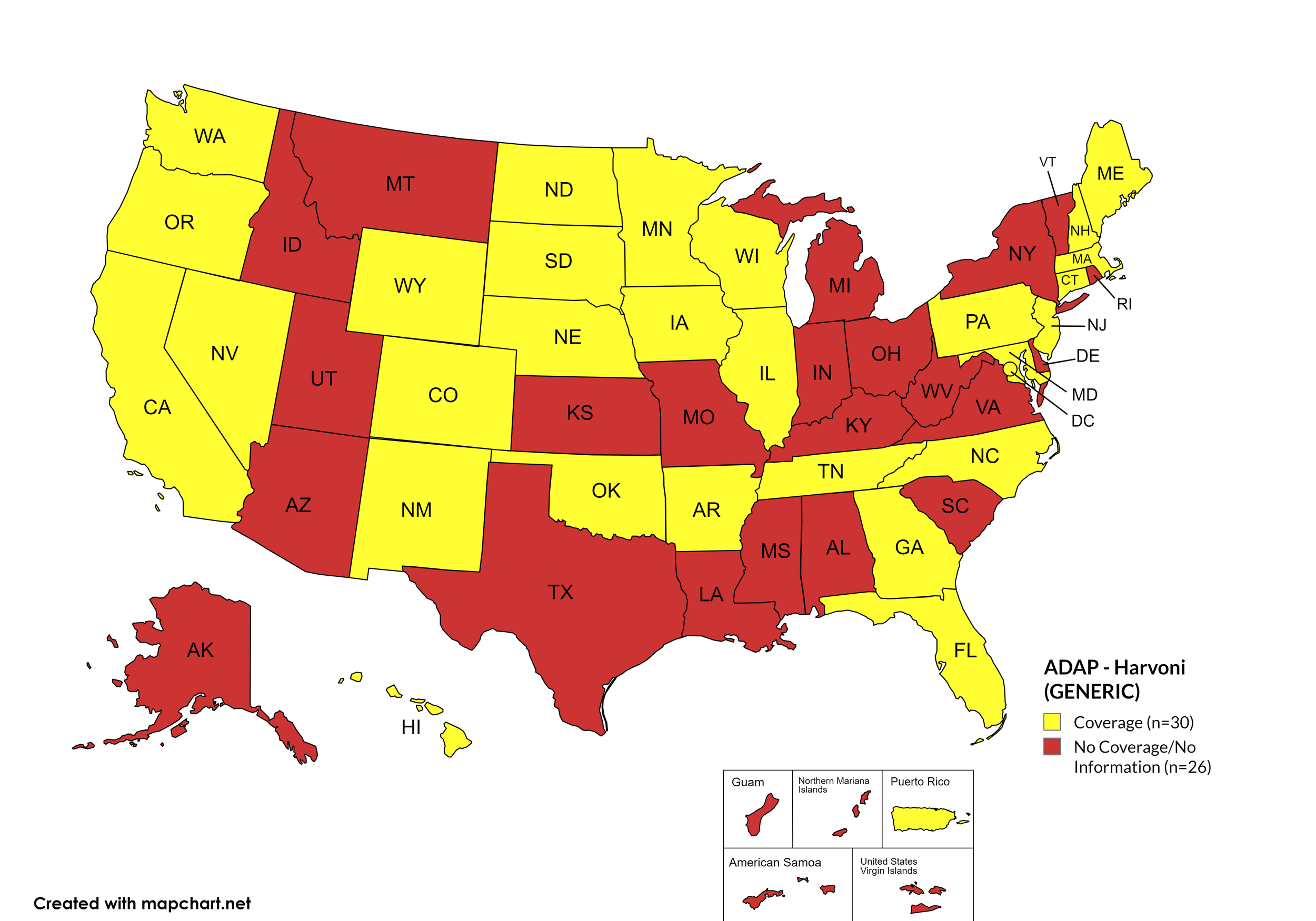
Harvoni (generic)
States with Harvoni (generic) Coverage: AR, CA, CO, CT, FL, GA, HI, IL, IA, ME, MD, MA, MN, NE, NV, NJ, NM, NC, ND, OK, OR, PA, SD, TN, WA, WI, WY, D.C.
States without Harvoni (generic)Coverage: AL, AK, AZ, DE, ID, IN, KS, KY, LA, MI, MS, MO, MT, NH, NY, OH, RI, SC, TX, UT, VT, VA, WV
Territories with Harvoni (generic) Coverage: P.R.
Figure 9. April 2026 ADAP Coverage - Harvoni (Generic)
Map Key: Yellow = Harvoni (Generic) Coverage; Red = No Harvoni (Generic) Coverage/No Information regarding Harvoni (Generic) Coverage
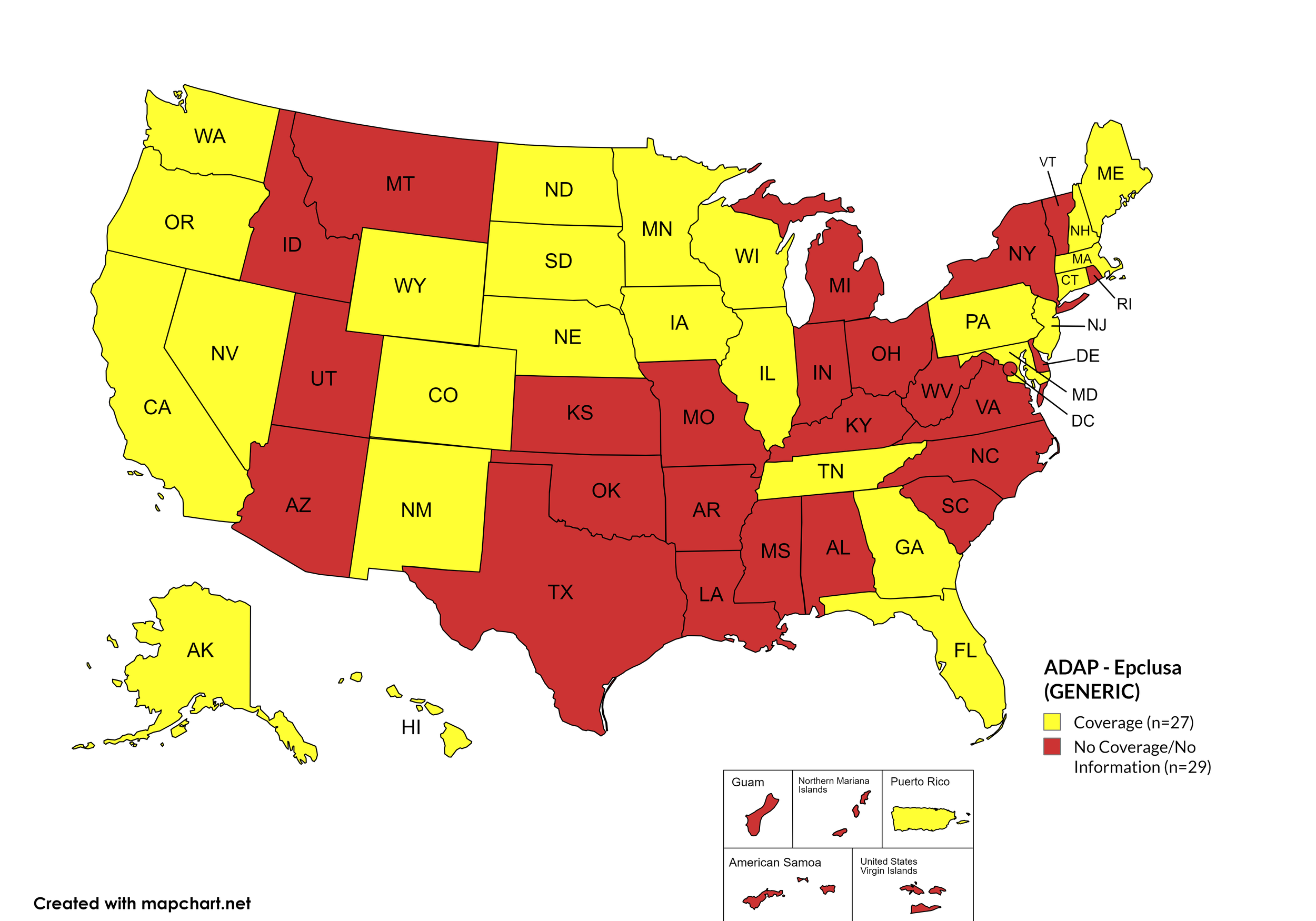
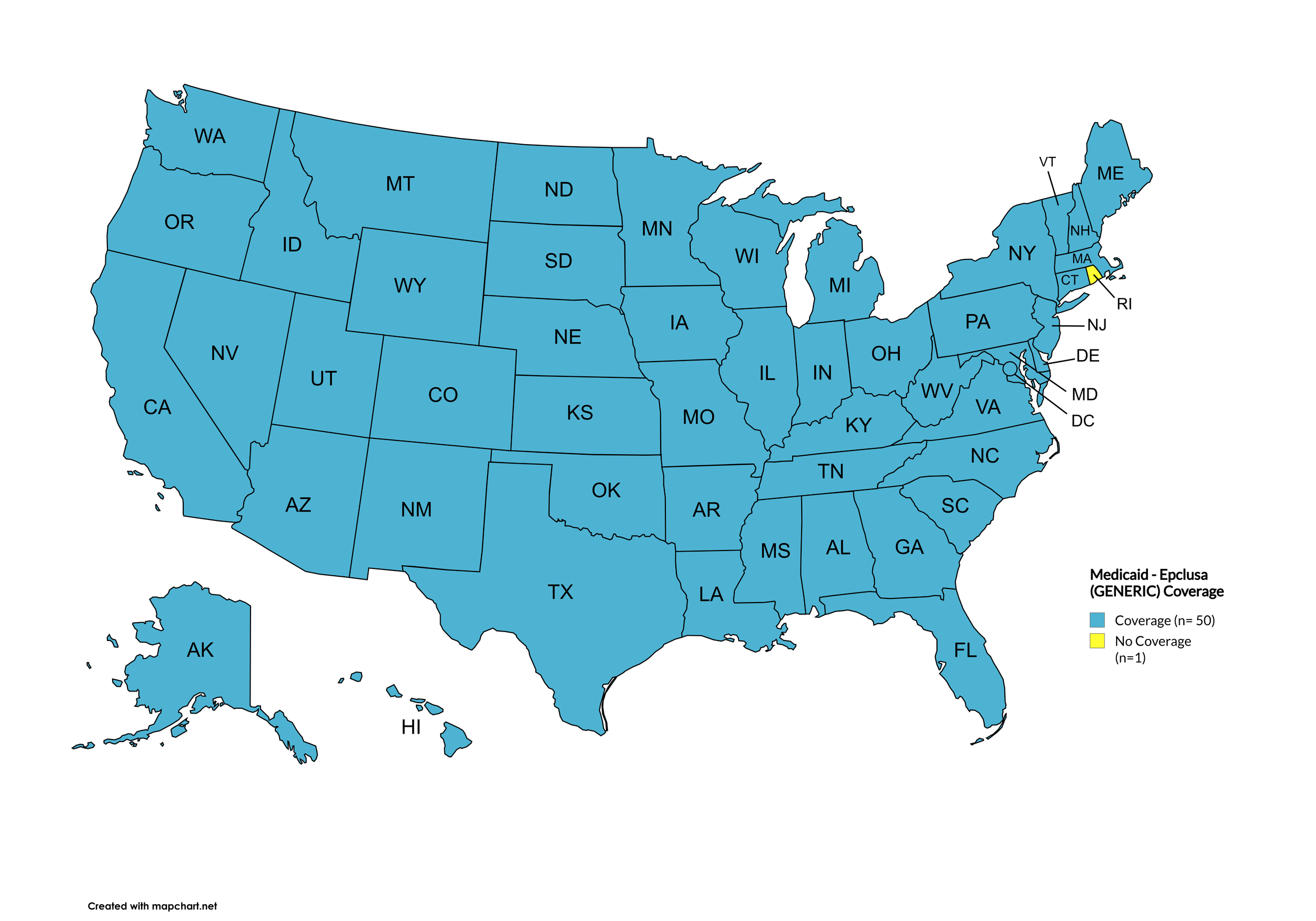
Epclusa (generic)
States with Epclusa (generic) Coverage: AR, CA, CO, CT, FL, GA, HI, IL, IA, ME, MD, MA, MN, NE, NH, NV, NJ, NM, ND, OR, PA, SD, TN, WA, WI, WY
States without Epclusa (generic) Coverage: AL, AK, AZ, DE, ID, IN, KS, KY, LA, MS, MO, MI, MT, NY, NC, OH, OK, RI, SC, TX, UT, VT, VA, WV, D.C.
Territories with Epclusa (generic) Coverage: P.R.
Figure 10. April 2026 ADAP Coverage - Epclusa (generic)
Map Key: Yellow = Epclusa (generic) Coverage; Red = No Epclusa (generic) Coverage/No Information regarding Epclusa (generic) Coverage
April 2026 Notes:
California now offers Basic Coverage (ribavirin)
States with Open Formularies: IL, IA, MA, MN, NE, NH, NJ, NM, ND, OH, OR, WA, WY
N.B. – Although Ohio is listed by NASTAD as having an open formulary, both NASTAD’s ADAP Formulary Database and Ohio’s ADAP website indicates that the state does not offer any treatment for HCV.
N.B. – Although North Dakota has adopted an open formulary, they provide only co-pay and deductible assistance for HCV medications.
N.B. – Wyoming's ADAP Open Formulary document, the following disclaimer related to HCV is made: Hepatitis C treatment medications (i.e. Harvoni, Sovaldi, Ribavirin, Zepatier, Epclusa) must be prior authorized. To be eligible, clients must have applied for prior authorization from their insurance plan and the WY ADAP Hepatitis C Treatment checklist must be completed and signed by the provider and client.
Colorado offers five coverage options – Standard ADAP, HIV Medical Assistance Program (HMAP), Bridging the Gap Colorado (BTGC), HIV Insurance Assistance Program (HIAP), and Supplemental Wrap Around Program (SWAP). 'Yes' indications in Figure 1 for Colorado denote that at least one of these programs offers coverage for each respective drug. Coverage through the Standard ADAP Formulary remains subject to funding availability.
On August 11th, 2023, Georgia's Department of Public Health issued a notice to Ryan White Part B District Coordinators, reading, in part, "Effective 8/14/2023, care providers will have the ability to order Hepatitis C medications for their eligible ADAP patients without the need for Prior Approval." Initially covered medications are limited to ribavirin, Zepatier, Mavyret, and generics for Epclusa and Harvoni.
Hawaii's ADAP notes the following: "Treatment slots for HCV direct-acting antivirals may be limited. Prescriber or pharmacy must call HDAP for slot."
Louisiana's ADAP (Louisiana Health Access Program – LA HAP) offers two coverage options – Uninsured (Louisiana Drug Assistance Program – L-DAP) and Insured (Health Insurance Program – HIP). HIP pays for the cost of treatment only if the client's primary insurance covers the drug under its formulary.
New Hampshire updated its formulary to require step therapy for Sovaldi, Harvoni, Zepatier, Epclusa, and Vosevi effective July 1, 2025. Preferred drugs are ribavirin, Pegasys, Mavyret, generic Harvoni, and generic Epclusa. This change affects coverage for brand-name DAAs, requiring patients to try preferred medications first.
Texas's ADAP maintains no HCV coverage, despite a brief period of covering DAAs in 2022.
Federal Legislative Development: The Cure Hepatitis C Act of 2025 (S.1941), introduced June 4, 2025, by Senators Bill Cassidy (R-LA) and Chris Van Hollen (D-MD), proposes establishing a national HCV elimination program with subscription-based drug access. NASTAD collaborated in drafting this bipartisan legislation, which could significantly expand treatment access if enacted.
NASTAD Database Status: The National Alliance of State and Territorial AIDS Directors formulary database remains current as of April 1, 2026, with no recent updates. Discrepancies between state websites and the database continue to create monitoring challenges.
3. MEDICAID PROGRAMS & HCV THERAPIES
All 50 states and the District of Columbia continue to offer some form of HCV coverage. All 50 States and the District of Columbia have expanded their Preferred Drug Lists to include at least one HCV Direct Acting Agent (DAA).
April 2026 Updates:
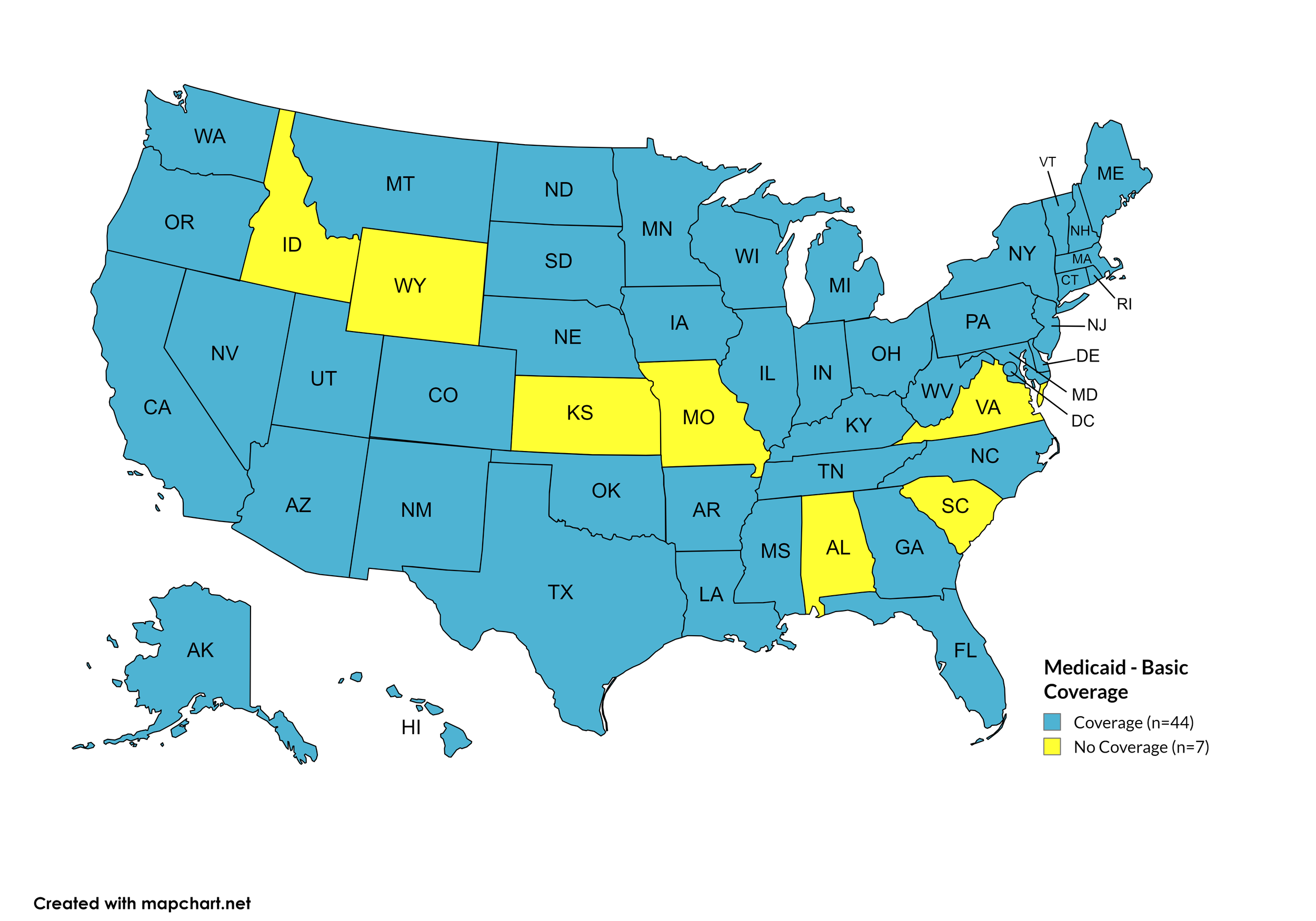
Basic Coverage
States with Basic HCV Medications Coverage: AK, AZ, AR, CA, CO, CT, DE, FL, GA, HI, IL, IN, IA, KY, LA, ME, MD, MA, MI, MN, MS, MT, NE, NV, NH, NJ, NM, NY, NC, ND, OH, OK, OR, PA, RI, SD, TN, TX, UT, VT, WA, WV, WI, WY, D.C.
States without Basic HCV Medications Coverage: AL, ID, KS, MO, SC, VA
Figure 11. April 2026 Medicaid Coverage - Basic HCV Medications
Map Key: Blue = Basic HCV Medication Coverage; Yellow = No Basic HCV Medication Coverage/No Information regarding Basic HCV Medication Coverage
Sovaldi
States with Sovaldi Coverage: AR, CA, CO, GA, HI, IL, KY, LA, MD, MA, MI, MN, MS, MO, MT, NE, NY, ND, PA, RI, SD, TX, UT, VT, WA, WI, D.C.
States without Sovaldi Coverage: AK, AL, AZ, CT, DE, FL, ID, IN, IA, KS, ME, NH, NJ, NM, NV, NC, OK, OH, OR, SC, TN, VA, WV, WY
Figure 12. April 2026 Medicaid Coverage - Sovaldi
Map Key: Blue = Sovaldi Coverage; Yellow = No Sovaldi Coverage/No Information regarding Sovaldi Coverage
Harvoni
States with Harvoni Coverage: AL, AR, CA, CO, GA, HI, IL, KY, LA, MI, MN, MS, MO, MT, NE, NY, ND, PA, RI, SD, TN, TX, UT, VT, WA, WV, WI, D.C.
States without Harvoni Coverage: AK, AZ, CT, DE, FL, IN, ID, IA, KS, ME, MD, MA, NH, NV, NJ, NM, NC, OK, OH, OR, SC, VA, WY
Figure 13. April 2026 Medicaid Coverage - Harvoni
Map Key: Blue = Harvoni Coverage; Yellow = No Harvoni Coverage/No Information regarding Harvoni Coverage
Zepatier
States with Zepatier Coverage: AL, AR, CA, CO, GA, HI, IL, IN, KY, LA, MD, MI, MN, MS, MO, MT, NE, NJ, NY, ND, OH, PA, SD, TX, UT, VT, WA, WI, WV, D.C.
States without Zepatier Coverage: AK, AZ, CT, DE, FL, ID, IA, KS, ME, MA, NH, NV, NM, NC, OK, OR, RI, SC, TN, VA, WY.
Figure 14. April 2026 Medicaid Coverage - Zepatier
Map Key: Blue = Zepatier Coverage; Yellow = No Zepatier Coverage/No Information regarding Zepatier Coverage
Epclusa
States with Epclusa Coverage: AL, CA, CO, GA, HI, IL, IN, KY, LA, MD, MI, MN, MO, MS, MT, NJ, NY, ND, PA, SC, SD, TN, TX, UT, VT, WA, WV, WI, D.C.
States without Epclusa Coverage: AK, AZ, AR, CT, DE, FL, ID, IA, KS, ME, MA, NE, NH, NV, NM, NC, OH, OK, OR, RI, VA, WY
Figure 15. April 2026 Medicaid Coverage - Epclusa
Map Key: Blue = Epclusa Coverage; Yellow = No Epclusa Coverage/No Information regarding Epclusa Coverage
Vosevi
States with Vosevi Coverage: AR, CA, CO, CT, FL, GA, HI, ID, IL, KY, LA, MD, MI, MN, MO, MS, MT, NE, NH, NY, NC, ND, OH, PA, SC, SD, TX, UT, VT, WA, WV, WI, D.C.
States without Vosevi Coverage: AK, AL, AZ, DE, IN, IA, KS, NH, NV, NJ, NM, ME, MA, OK, OR, RI, TN, VA, WY
Figure 16. April 2026 Medicaid Coverage - Vosevi
Map Key: Blue = Vosevi Coverage; Yellow = No Vosevi Coverage/No Information regarding Vosevi Coverage
Mavyret
States with Mavyret Coverage: AL, AK, AZ, AR, CA, CO, CT, DE, FL, GA, HI, ID, IL, IN, IA, KS, KY, LA, ME, MD, MA, MI, MN, MS, MO, MT, NE, NV, NH, NJ, NM, NY, NC, ND, OH, OK, OR, PA, RI, SC, SD, TN, TX, UT, VT, VA, WA, WV, WI, WY, D.C.
Figure 17. April 2026 Medicaid Coverage - Mavyret
Map Key: Blue = Mavyret Coverage; Yellow = No Mavyret Coverage/No Information regarding Mavyret Coverage
Pegasys
States with Pegasys Coverage: AK, AZ, AR, CA, CT, FL, GA, HI, IL, IN, IA, KY, LA, ME, MD, MI, MN, MS, MT, NE, NV, NH, NJ, NM, NC, OH, OK, OR, PA, RI, SD, TN, TX, VT, WA, WV, WI, D.C.
States without Pegasys Coverage: AL, CO, DE, ID, KS, MA, MO, NY, ND, SC, UT, VA, WY
Figure 18. April 2026 Medicaid Coverage - Pegasys
Map Key: Blue = Pegasys Coverage; Yellow = No Pegasys Coverage/No Information regarding Pegasys Coverage
Harvoni (generic)
States with Harvoni (generic) Coverage: AL, AR, CA, CO, DE, GA, HI, IL, KY, LA, MD, MA, MI, MN, MO, MS, MT, NE, NH, ND, OK, PA, RI, SD, TN, TX, UT, VT, WA, WV, WI, D.C.
States without Harvoni (generic) Coverage: AK, AZ, CT, FL, ID, IN, IA, KS, ME, NM, NJ, NV, NC, OK, OH, OR, SC, VA, WY
Figure 19. April 2026 Medicaid Coverage - Harvoni (generic)
Map Key: Blue = Harvoni (generic) Coverage; Yellow = No Harvoni (generic) Coverage/No Information regarding Harvoni (generic) Coverage
Epclusa (generic)
States with Epclusa (generic) Coverage: AK, AL, AZ, AR, CA, CO, CT, DE, FL, GA, HI, ID, IL, IN, IA, KS, KY, LA, ME, MD, MA, MI, MN, MS, MO, MT, NE, NV, NH, NJ, NM, NY, NC, ND, OK, OH, OR, PA, SC, SD, TN, TX, UT, VT, VA, WA, WV, WI, WY, D.C.
States without Epclusa (generic) Coverage: RI
Figure 20. April 2026 Medicaid Coverage - Epclusa (generic)
Map Key: Blue = Epclusa (generic) Coverage; Yellow = No Epclusa (generic) Coverage/No Information regarding Epclusa (generic) Coverage
April 2026 Notes:
The following states’ Medicaid programs offer multiple coverage plans for their respective Medicaid clients. The plan highlighted in bold typeface represents the most comprehensive plan with the most drugs covered in the respective state:
Hawaii – (1.) Alohacare Advantage Plus; (2.) HMSA; (3.) Kaiser Permanente; (4.) UnitedHealthcare QUEST Integration; (5.) WellCare
New Jersey – (1.) Aetna; (2.) AmeriGroup NJ (Now Wellpoint); (3.) Horizon NJ Health; (4.) UnitedHealthcare of New Jersey (New Jersey Family Care);
New Mexico – (1.) BlueCross BlueShield of New Mexico; (2.) Presbyterian Centennial Care; (3) Western Sky Community Care
Kentucky has a Unified Medicaid Formulary
Louisiana has a Unified Medicaid Formulary
Ohio – Ohio has a Unified Medicaid Formulary that applies to all MCOs
No data has been made available by the Medicaid programs in the U.S. Territories.
California has gone back to covering Vosevi. It had stopped coverage during a previous Watch period.
Indiana - Harvoni, Sovaldi, Vosevi, and generic Harvoni are now non-preferred, requiring failure of preferred agents plus prior authorization
New Hampshire - Epclusa, Harvoni, Sovaldi, Vosevi, and Zepatier are now non-preferred, requiring failure of preferred options before allowing use
Major Policy Changes That Occurred (April-October 2025):
Hawaii Alohacare Advantage Plus plan now covers Pegasys and generic Epclusa. Kaiser Permanente plan now covers generic Harvoni and generic Epclusa.
Louisiana removed prior authorization requirements for generic HCV medications, significantly improving access to sofosbuvir/velpatasvir (generic Epclusa) and ledipasvir/sofosbuvir (generic Harvoni).
Maryland changed Harvoni from preferred to non-preferred status requiring prior authorization, while Mavyret is now preferred with no prior authorization required.
Nevada substantially reduced its HCV formulary by removing Sovaldi, Harvoni, Zepatier, Vosevi, and generic Harvoni, leaving only ribavirin, Pegasys, generic Epclusa, and Mavyret as treatment options.
North Dakota removed prior authorization requirements for Mavyret.
Ohio Medicaid began offering HCV treatment for the first time, with formulary options including ribavirin, Mavyret, Pegasys, and generic Epclusa.
South Carolina added Epclusa to its preferred drug list.
Continuing Policies:
All state Medicaid programs have removed fibrosis restrictions for initial treatment.
There are currently no states that require sobriety as a prerequisite for hepatitis C treatment.
For clarification, if a state requires a complicated prior authorization process or step therapy requiring failure on preferred medications, that is considered 'no coverage'.
Rhode Island remains the only state that does not cover generic Epclusa.
*Medicaid coverage excludes patients from most drug manufacturer patient assistance programs (PAPs)
4. VETERANS PROGRAMS & HCV THERAPIES
The Veteran's Administration (VA) currently offers coverage for all HCV drugs. This is according to the most recent VA National Formulary, dated October 2025 (U.S. Dept. of V.A., 2025a). The VA Treatment Considerations and Choice of Regimen for HCV-Mono-Infected and HIV/HCV Co-Infected Patients, dated March 2021 (U.S. Dept. of V.A., 2021b) lists the following therapies as preferred treatments:
Abbreviations:
- CTP – Child-Turcotte-Pugh (score used to assess severity of cirrhosis)
- IU/mL – International Units Per Milliliter
- PEG-IFN/IFN – Peginterferon/Interferon
- RAS – Resistance-associated substitutions
Genotype 1:
Treatment-naïve without or with cirrhosis (CTP A):
Pangenotypic regimens
Mavyret: 3 tablets orally daily with food for 8 weeks; may consider 12 weeks in patients with poor prognostic factors
Epclusa: 1 tablet orally daily for 12 weeks
Non-pangenotypic regimens:
Zepatier: 1 tablet orally daily for 12 weeks if GT1a without baseline NS5A RAS or GT1b
Harvoni: 1 tablet orally daily
If HCV-noninfected, non-cirrhotic, and HCV RNA baseline <6 million IU/mL: 8 weeks
If cirrhotic, baseline HCV RNA ≥6 million IU/mL, HIV/HCV-co-infected, or African American: 12 weeks
Consider adding ribavirin in CTP A patients
Treatment-naïve with decompensated cirrhosis (CTP B or C):
Harvoni: 1 tablet orally daily + ribavirin (600 mg/day and increase by 200 mg/day every 2 weeks only as tolerated) for 12 weeks
Epclusa: 1 tablet orally daily + ribavirin (1000 mg/day - <75kg – or 1,200 mg daily - ≥75kg – orally daily in 2 divided doses with food) for 12 weeks; start at lower ribavirin doses as clinically indicated (e.g., baseline Hgb).
Treatment-experienced (NS5A- and SOF-naïve [e.g., failed PEG-IFN/RBV ± NS3/4A PI]) without or with cirrhosis (CTP A)
Pangenotypic regimens:
Mavyret: 3 tablets orally daily with food
If PEG-IFN/RBV-experienced: 8 weeks if non-cirrhotic or 12 weeks if cirrhotic
If NS3/4A PI + PEG-IFN/RBV-experienced: 12 weeks
Vosevi: 1 tablet orally daily for 12 weeks
Non-pangenotypic regimens
Zepatier: 1 tablet orally daily for 12 weeks if GT1b, or if failed only PEG-IFN/RBV and GT1a without baseline NS5A RAS
Harvoni: 1 tablet orally daily for 12 weeks
Treatment-experienced (NS5A-naïve and SOF-experienced) without or with cirrhosis (CTP A)
Mavyret: 3 tablets orally daily with food
If PEG-IFN/RBV + Sovaldi-experienced: 8 weeks if non-cirrhotic or 12 weeks if cirrhotic
If Olysio + Sovaldi-experienced: 12 weeks
Epclusa: 1 tablet orally daily for 12 weeks if GT1b
Vosevi: 1 tablet orally daily with food for 12 weeks if GT1a
Treatment-experienced (prior NS5A-containing regimen) without or with cirrhosis (CTP A)
Mavyret: 3 tablets orally daily with food for 16 weeks if failed only an NS5A inhibitor without NS3/4A PI (e.g., Harvoni)
Vosevi: 1 tablet orally daily with food for 12 weeks
Treatment-experienced with decompensated cirrhosis (CTP B or C)
Epclusa: 1 tablet orally daily + RBV; start at lower RBV doses as clinically indicated (e.g., baseline Hgb);
If NS5A-naïve: 12 weeks
If NS5A-experienced: 24 weeks; NOT FDA approved for 24 weeks
Genotype 2:
Treatment-naïve or treatment-experienced (PEG-IFN/IFN ± RBV or Sovaldi + RBV ± PEG-IFN) without or with cirrhosis (CTP A)
Mavyret: 3 tablets orally daily with food for 8 weeks; 12 weeks if CTP A and treatment-experienced or in patients with poor prognostic factors
Epclusa: 1 tablet orally daily for 12 weeks
Treatment-experienced (NS5A-experienced) without or with cirrhosis (CTP A)
Vosevi: 1 tablet orally daily with food for 12 weeks
Treatment-naïve or treatment-experienced patients with decompensated cirrhosis (CTP B or CTP C)
Epclusa: 1 tablet orally daily + ribavirin; start at lower ribavirin doses as clinically indicated (e.g., baseline Hgb)
If NS5A-naïve: 12 weeks
If NS5A-experienced: 24 weeks
Genotype 3:
Treatment-naïve without cirrhosis or with cirrhosis (CTP A)
Mavyret: 3 tablets orally daily with food for 8 weeks; may consider 12 weeks if cirrhotic or in patients with poor prognostic factors
Epclusa: 1 tablet orally daily for 12 weeks
If CTP A, test for NS5A RAS
Add ribavirin if Y93H RAS present
Treatment-experienced (PEG-IFN ± RBV or Sovaldi + RBV ± PEG-IFN) without or with cirrhosis (CTP A)
Mavyret: 3 tablets orally daily with food for 16 weeks
Treatment-experienced (NS5A-experienced) without or with cirrhosis (CTP A)
Vosevi: 1 tablet orally daily with food for 12 weeks
If CTP A, consider adding ribavirin (no supporting data)
Treatment-naïve or treatment-experienced with decompensated cirrhosis (CTP B or CTP C)
Epclusa: 1 tablet orally daily + ribavirin; start at lower ribavirin doses as clinically indicated (e.g., baseline Hgb)
If NS5A-naïve: 12 weeks
If NS5A-experienced: 24 weeks
Genotype 4:
Treatment-naïve without or with cirrhosis (CTP A)
Pangenotypic regimens
Mavyret: 3 tablets orally daily with food for 8 weeks; may consider 12 weeks in patients with poor prognostic factors
Epclusa: 1 tablet orally daily for 12 weeks
Non-pangenotypic regimens
Zepatier: 1 tablet orally daily for 12 weeks
Harvoni: 1 tablet orally daily for 12 weeks
Treatment-naïve with decompensated cirrhosis (CTP B or C)
Pangenotypic regimen
Epclusa: 1 tablet orally daily + RBV for 12 weeks; start at lower ribavirin doses as clinically indicated (e.g., baseline Hgb)
Non-pangenotypic regimen:
Harvoni: 1 tablet orally daily + ribavirin (600 mg/day and increase by 200 mg/day every 2 weeks only as tolerated) for 12 weeks
Treatment-experienced (Sovaldi-experienced and NS5A-naïve) without or with cirrhosis (CTP A)
Mavyret: 3 tablets orally daily with food for 8 weeks if NS3/4A PI-naïve without cirrhosis, and 12 weeks if NS3/4A PI-experienced or CTP A
Epclusa: 1 tablet orally daily + ribavirin for 12 weeks; start at lower ribavirin doses as clinically indicated (e.g., baseline Hgb)
Treatment-experienced (NS5A-experienced) without or with cirrhosis (CTP A)
Vosevi: 1 tablet orally daily with food for 12 weeks
Treatment-experienced with decompensated cirrhosis (CTP B or CTP C)
Epclusa: 1 tablet orally daily + ribavirin; start at lower ribavirin doses as clinically indicated (e.g., baseline Hgb)
If NS5A-naïve: 12 weeks
If NS5A-experienced: 24 weeks; NOT FDA approved for 24 weeks
April 2026 Notes:
Program Stability: The VA HCV treatment program maintained comprehensive coverage throughout the August-October 2025 period with a few formulary restrictions, coverage limitations, or policy changes implemented.
Coverage Status: Direct-acting antivirals (DAAs) remain available including:
Sovaldi (sofosbuvir) - is no longer on the national formulary and requires a non-formulary drug request and prior approval to be dispensed.
Harvoni (ledipasvir/sofosbuvir) and generic formulations
Zepatier (elbasvir/grazoprevir) - is no longer on the national formulary and requires a non-formulary drug request and prior approval to be dispensed. It also has very specified criteria for its use.
Epclusa (sofosbuvir/velpatasvir) and generic formulations
Vosevi (sofosbuvir/velpatasvir/voxilaprevir)
Mavyret (glecaprevir/pibrentasvir)
Ribavirin and Pegasys for combination therapy (Ribavirin-free regimens are preferred)
Screening and Treatment Success: The VA continues to demonstrate exceptional performance in HCV care with 75% of veterans in care tested for HCV and over 95% of antibody-positive veterans receiving confirmatory testing. Patients with successful treatment who become re-infected are eligible for retreatment.
Formulary Updates: The most recent VA National Formulary update occurred February 2026, confirming continued availability of all HCV medications with several restrictions.
Treatment Guidelines: The VA Treatment Considerations and Choice of Regimen guidelines from March 2021 remain current, providing evidence-based treatment recommendations across all HCV genotypes and clinical scenarios.
Comparison to Other Programs: The VA's stable, comprehensive coverage contrasts significantly with the variability and restrictions seen in state ADAP and Medicaid programs, demonstrating the benefits of integrated healthcare system management.
5. PATIENT ASSISTANCE PROGRAMS
The drug manufacturers and various national nonprofit organizations offer a variation of patient assistance programs (PAPs) to assist patients in accessing treatments. They include:
Support Path (Gilead Sciences):
Status: ACTIVE
Financial Assistance
Provides Co-Pay Coupons for Sovaldi, Harvoni, Harvoni (Generic), Epclusa, Epclusa (Generic), and Vosevi
Patients without insurance may be eligible to recieve medication free of charge through the Support Path Patient Assistance Program (PAP)
Co-Pay Coupons cover out-of-pocket costs up to 25% of the catalog price of a 12-week regimen (3 bottles/packages) of Sovaldi, Harvoni, Harvoni (Generic), Epclusa, Epclusa (Generic), or Vosevi
Excludes patients enrolled in Medicare Part D, Medicaid, TRICARE, VA, DOD, or Puerto Rico Government Health Insurance Plan
Gilead may reduce or discontinue the cost-sharing assistance available if it is discovered that a patient’s insurer implements co-pay maximizer or co-pay accumulator programs.
MAJOR CHANGE: Effective May 5, 2025, transitioned from retail pharmacy to a mail-order delivery system
MAJOR CHANGE: Discontinued providing free medications for Sovaldi and several HIV medications due to generic availability
Insurance Support
Researches and verifies patients’ benefits, and gives information they need about coverage options and policies
Explains Prior Authorization process and works with HCV Specialist's office so they can submit PA forms to a patient's insurance company
May be able to provide assistance with appeals process
Special Exceptions Ended
DISCONTINUED: Special exceptions for Truvada PrEP for individuals assigned female at birth ended July 31, 2025
Website: http://www.mysupportpath.com/
AbbVie Mavyret Co-Pay Savings Card:
Status: ACTIVE
Financial Assistance
Patient may be eligible to pay as little as $5
Excludes patients enrolled in Medicare Part D, Medicare Advantage, Medigap, Medicaid, TRICARE, Department of Defense, or Veterans Affairs programs
NeedyMeds:
Status: ACTIVE
NeedyMeds Drug Discount Card
Designed to lower cost of prescription medications by up to 80% at participating pharmacies
Price finder tool for the drug discount card
No eligibility requirements
CANNOT be used in combination with government healthcare programs, but CAN be used IN PLACE of program
CANNOT be combined with other offers
Website: https://www.needymeds.org/drug-discount-card Search for other assistance programs: http://ow.ly/fEJo309cJ7Z
The Assistance Fund:
Status: WAITLISTED (accepting patients for waitlist)
Requires provider referral
Copay assistance
Eligibility Criteria:
US citizen or permanent resident
Diagnosed with the disease for which you are applying
Prescribed an FDA-approved treatment for the disease
Have prescription coverage for the prescribed treatment
Meet financial eligibility criteria based upon household income and size
At the end of each calendar year, waitlist applications expire. Patients still seeking assistance must join waitlists for the subsequent calendar year.
Patient Advocate Foundation Co-Pay Relief:
Status: OPEN
Maximum award of $6,000 per year
Eligibility Requirements:
Patient must be insured, and insurance must cover prescribed medication
Confirmed HCV diagnosis
Reside and receive treatment in the U.S.
Income falls at 500% or less of FPG (Federal Poverty Guideline) with consideration of the Cost of Living Index (COLI) and the number in the household
Patient Access Network (PAN) Foundation:
Status: CLOSED (not currently accepting applications for new or renewal patients but eligible patients can join the waitlist here or call 1-866-316-7263)
Co-Pay Assistance with a maximum award of $3,800
PROGRAM ADJUSTMENT: Beginning January 1, 2025, adjusted grant amounts in response to Medicare Part D's new $2,000 out-of-pocket cap, maintaining goal to cover 100% of costs for most patients
Patients may apply for additional assistance during their eligibility period, subject to availability of funding
Eligibility Requirements:
Must be getting treatment for HCV
Have insurance that covers prescribed HCV medication
Medication must be listed on PAN's list of covered medications: https://www.panfoundation.org/index.php/en/patients/medications-covered
Income falls below 500% of FPL
Residing and receiving treatment in the U.S. (citizenship NOT required)
Website: https://www.panfoundation.org/disease-funds/hepatitis-c/
HealthWell Foundation:
Status: CLOSED
Co-Pay Assistance with a maximum award of $10,000
Minimum Co-Pay Reimbursement Amount: None
Minimum Premium Reimbursement Amount: None
Fund reopened in April 2025 after previous closure due to insufficient funding and has closed again to new applicants due to insufficient funding. All current grantees’ grants will remain active for the entire 12 month grant cycle or until you have exhausted your allocated grant amount, whichever comes first.
Eligibility Requirements:
Must be being treated for HCV
Have insurance that covers HCV prescribed medication
Income falls below 500% of FPL
Receiving treatment in the U.S.
Website: https://www.healthwellfoundation.org/fund/hepatitis-c/
April 2026 Notes:
Major Program Changes:
Gilead Support Path Program underwent significant restructuring on May 5, 2025:
Transitioned from retail pharmacy distribution to mail order delivery system
Discontinued providing free medications for Sovaldi and several HIV medications due to widespread generic availability
Ended special exceptions for Truvada PrEP for individuals assigned female at birth effective July 31, 2025
Maintained co-pay assistance for hepatitis C medications
PAN Foundation adapted to new Medicare Part D regulations by adjusting grant amounts beginning January 1, 2025, to work within the new $2,000 annual out-of-pocket cap while maintaining comprehensive cost coverage for eligible patients.
Patient Advocate Foundation Co-Pay Relief has reopened its hepatitis C program
The Assistance Fund moved to waitlist status, indicating funding constraints but continued program operation for future applicants.
HealthWell Foundation remains the most significant positive development, maintaining its April 2025 reopening with the highest award amount ($30,000) among active programs. (UPDATE…program is still closed)
Impact Assessment: The closure and modification of multiple PAPs reflects the evolving treatment landscape as generic HCV medications become more affordable and accessible. However, these changes may create access gaps for patients who don't qualify for remaining programs or whose insurance doesn't adequately cover newer generic formulations.
Current Active Programs: Support Path, AbbVie, NeedyMeds, Patient Advocate Foundation and The Assistance Fund waitlist
Closed Programs: PAN Foundation, HealthWell Foundation
6. HARM REDUCTION PROGRAMS
Harm Reduction, as it relates to opioid abuse and HCV, are measures designed to serve as preventive or monitoring efforts in combating opioid prescription drug and heroin abuse, and as an effect, helping to prevent the spread of HCV and HIV. The Co-Infection Watch covers the following measures: Syringe Exchange, Expanded Naloxone Access, State Authorized Safe Consumption Sites, Updated Paraphernalia Laws (allowing for possession of substance testing strips), Good Samaritan Laws, Required Prescriber Education. (Editor’s Note: Program descriptions provided herein).
April 2026 Updates:
Syringe Exchange
Syringe Services Programs (SSPs) exist to provide injection drug users (or those whose prescriptions require injection) with clean syringes and/or in exchange for used ones. (N.b. – states listed as "at least one SSP…” indicate only that a Syringe Services Program (SSP) exists within the state, regardless of the legality of SSPs under state law).
States with Syringe Exchange: AK, AZ, AR, CA, CO, CT, DE, FL, GA, HI, IL, IN, IA, KY, LA, ME, MD, MA, MI, MN, MO, MT, NV, NH, NJ, NM, NY, NC, ND, OH, OK, OR, PA, RI, SC, TN, TX, UT, VT, VA, WA, WV, WI, D.C.
States without Syringe Exchange: AL, ID, KS, MS, NE, SD, WY
Territories with Syringe Exchange: Puerto Rico, U.S. Virgin Islands
Figure 21. April 2026 Syringe Exchange Coverage
Map Key: Purple = Syringe Exchange(s); Red = No Syringe Exchange(s); Grey = No Information
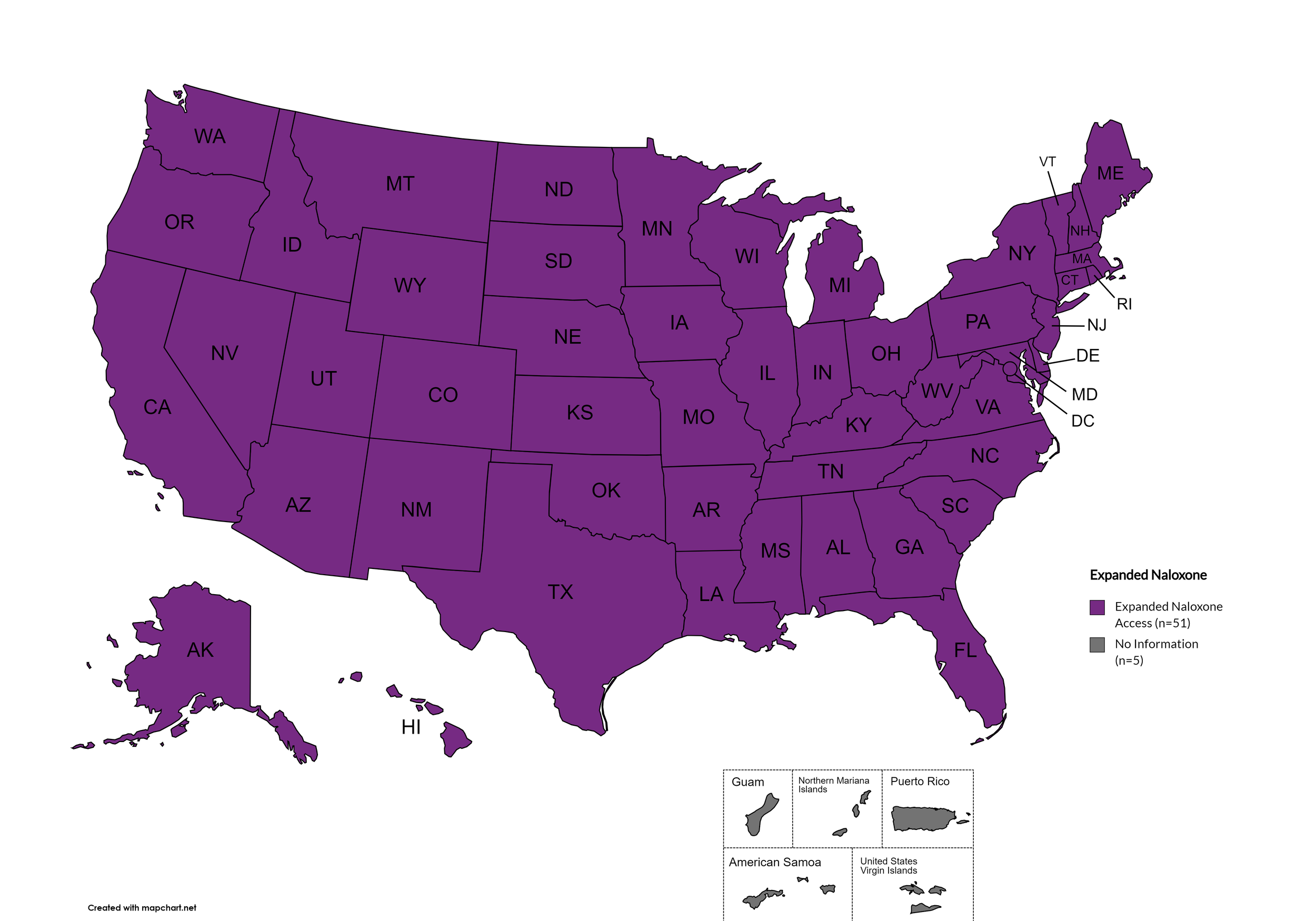
Expanded Naloxone
Naloxone is a drug used to counteract the effects of opioid overdoses. Expanded Access refers to having statutes or state standing orders in place that allow pharmacies to dispense naloxone without a prescription. This means those in danger of overdose, those who are caregivers for them, or anyone who may come in contact with those in danger of overdose can walk into a pharmacy and obtain naloxone without a prescription. Removing the requirement of a patient-doctor relationship via prescription enhances access.
States with Expanded Naloxone: AL, AK, AZ, AR, CA, CO, CT, DE, FL, GA, HI, ID, IL, IN, IA, KS, KY, LA, ME, MD, MA, MI, MN, MO, MS, MT, NE, NV, NH, NJ, NM, NY, NC, ND, OH, OK, OR, PA, RI, SC, SD, TN, TX, UT, VT, VA, WA, WV, WI, WY, D.C.
States without Expanded Naloxone: None
Territories with Expanded Naloxone: Unknown
Figure 22. April 2026 Expanded Naloxone Coverage
Map Key: Purple = Expanded Naloxone; Red = Restricted Naloxone; Gray = No Information
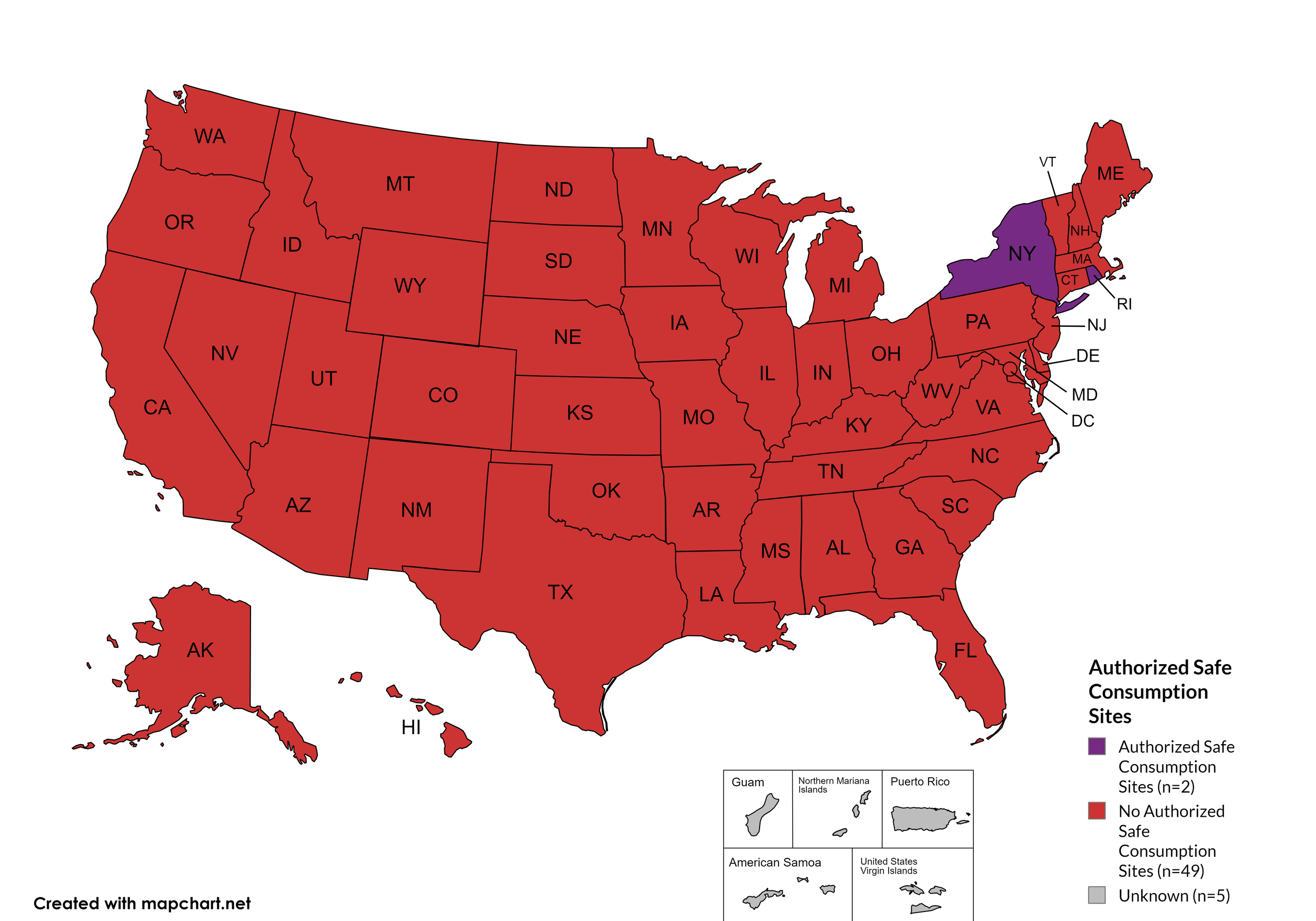
State Authorized Safe Consumption Sites
Federal law prohibits the distribution, possession, and consumption of certain controlled substances. Safe Consumption Sites (SCSs) exist to provide injection drug users (or those whose prescriptions require injection) with clean syringes and/or in exchange for used ones, offer wound care supplies, allow for injection drug users to consume drugs, offer infectious disease screening, and other linkage to care opportunities. This section monitors state authorized safe consumption site programs and pilot projects related to safe consumption sites.
States with Safe Consumption Sites: NY, RI
States without Safe Consumption Sites: AL, AK, AZ, AR, CA, CO, CT, DE, FL, GA, HI, ID, IL, IN, IA, KS, KY, LA, ME, MD, MA, MI, MN, MO, MS, MT, NE, NV, NH, NJ, NM, NC, ND, OH, OK, OR, PA, SC, SD, TN, TX, UT, VT, VA, WA, WV, WI, WY, D.C.
Territories with Safe Consumption Sites: None
Figure 23. April 2026 State Authorized Safe Consumption Sites
Map Key: Purple = States with sites; Red = States without sites
Updated Paraphernalia Laws
State paraphernalia laws have long prohibited possession of certain drugs use related materials, including harm reduction materials like fentanyl testing strips. Some states have modernized their criminal codes to allow for possession of testing strips and may also have health department programs distributing testing strips.
States with Updated Paraphernalia Laws: AL, AK, AZ, AR, CA, CO, CT, DE, FL, GA, HI, ID, IL, IN, KS, KY, LA, MD, MA, ME, MI, MN, MO, MS, MT, NE, NV, NH, NJ, NM, NY, NC, ND, OH, OK, OR, PA, RI, SC, SD, TN, UT, VT, VA, WA, WV, WI, WY, D.C.
States without Updated Paraphernalia Laws: IA
Territories with Updated Paraphernalia Laws: Unknown
Figure 24. April 2026 Updated Paraphernalia laws
Map Key: Red = Without Updated Laws; Purple = With Updated Laws; Gray = No Information
Expanded Good Samaritan Laws
Expanded Good Samaritan Laws are laws that are designed to protect persons seeking emergency services for drug overdoses from drug-related charges or prosecutions, regardless of possession or consumption of illegal, illicit substances, or drug paraphernalia. Good Samaritan laws may or may not provide protection to those currently under parole or probation. Good Samaritan laws listed do NOT prohibit arrest.
States with Expanded Good Samaritan Laws: AZ, CA, CT, DE, Fl, GA, HI, ID, IL, KY, LA, MD, MN, MS, MO, MT, NB, NV NJ, NM, NY, ND, PA, RI, TN
States without Expanded Good Samaritan Laws: AL, AK, AR, CO, IN, IA, KS, ME, MA, MI, NH, NC, OH, OK, OR, SC, SD, TX, UT, VT, VA, WV, WI, WY
Territories with Expanded Good Samaritan Laws: Unknown
Figure 25. April 2026 Good Samaritan Laws Coverage
Map Key: Purple = Good Samaritan Laws; Red = No Good Samaritan Laws; Gray: No Information
Prescriber Education Required
States that require/do not require through legislative action or regulatory or licensing bodies that prescribing physicians undergo special training in addition to or as part of their initial education to become prescribers related to safer controlled substance and/or pain management prescribing and utilization practices.
States with Prescriber Education Required: AL, AK, AZ, AR, CA, CO, CT, DE, FL, GA, HI, ID, IL, IN, IA, KS, KY, LA, ME, MD, MA, MI, MO, MN, MS, NE, NV, NH, NJ, NM, NY, NC, ND, OH, OK, OR, PA, RI, SC, TN, TX, UT, VT, VA, WA, WV, WI, WY, D.C.
States without Prescriber Education Required: MT, SD
Territories with Prescriber Education Required: Unknown
Figure 26. April 2026 Prescriber Education Required Coverage
Map Key: Purple = Prescriber Ed Required; Red = No Prescriber Ed Required; Gray = No Information
April 2026 Notes:
Metrics for Mandatory PDMP reporting, Doctor Shopping Laws, Physical Exam, ID Requirements, and Lock-in Pharmacy programs have been permanently deleted from the Watch due to redundancy or outdatedness.
Metric definition for Expanded Naloxone was updated. Medicaid covers naloxone in all states. However, even though naloxone was previously made available OTC federally, some states still required a prescription for it at the pharmacy. Expanded Naloxone designation also addresses issues such as its availability in schools and other public places, the state-by-state variation in who other than pharmacists is allowed to dispense naloxone, and more.
Added metrics for monitoring state paraphernalia laws regarding possession of testing strips and state authorized safe consumption sites, including legislatively authorized pilot projects.
NC, ND, and VT still have controlled substance testing equipment on their drug paraphernalia law statues but provide a carve-out to allow testing strips.
Metric definition for Prescriber Education has been updated to exclude “recommended” and only reflect those states which have laws or licensing board requirements of initial and/or continuing education for prescribers with regard to pain management and/or the prescription of controlled substances.
Some states have general requirements regarding “controlled substances”, some states are explicit with regard to category of controlled substance or type of controlled substance (ie. “opioids”).
This adjustment clarifies that MT and SD are the only states that do not require opioid specific and/or pain management specific and/or controlled substances prescribing education by law or licensing institution in either core or continuing education for providers.
This adjustment clarifies that KS, MO, and ND do require opioid specific and/or pain management specific and/or controlled substances prescribing education by law or licensing institution in either core or continuing education for providers.
The immunity provided under Good Samaritan Laws only applies when there are personal usage amounts of drugs present. It does not apply when there are distribution-level amounts.
In March 2024, Idaho repealed its five-year-old Syringe and Needle Act by passing House Bill 617. The governor signed it into law for it to go into effect July 1, 2024.
Idaho's legalization of fentanyl strips officially went into effect July 1, 2024.
Indiana passed House Bill 1167 to decriminalize fentanyl testing strips, signed by Governor Mike Braun on April 10, 2025, effective July 1, 2025. This removes Indiana from states without updated paraphernalia laws.
Iowa remains the only state that has not updated its paraphernalia laws to allow possession of fentanyl testing strips for harm reduction purposes.
Nebraska's standing order for Naloxone was extended beyond its original August 2024 expiration date.
Rhode Island's first safe consumption site opened in December 2024 at 45 Willard Avenue, next to the Rhode Island Hospital Campus in Providence. The site has served over 500 visitors and prevented 27 overdose deaths in its first six months of operation. The legislature approved a two-year extension of the pilot program in April 2025, extending authorization through March 2026.
Wyoming law does not explicitly outlaw fentanyl testing strips, however they have not been widely distributed.
The DEA's COVID-19-based waiver of in-person exams that was extended through December 31, 2024, has now been extended again through December 31, 2025, as announced in the "Third Temporary Extension of COVID-19 Telemedicine Flexibilities for Prescription of Controlled Medications." The DEA and HHS stated they "continue to carefully consider the input received and are working to promulgate a final set of telemedicine regulations" while extending the current flexibilities.
Additionally, on January 16, 2025, the DEA announced "three new rules to make permanent some temporary telemedicine flexibilities established during the COVID-19 public health emergency while also establishing new patient protections." These proposed rules, currently open for public comment, would create special registration categories for telemedicine prescribing after the temporary extension expires.
State Developments:
California launched a groundbreaking direct-to-consumer naloxone program on April 21, 2025, offering CalRx naloxone at $24 per twin-pack—nearly 50% below standard market prices.
New Hampshire distributed over 58,000 naloxone kits statewide and became the first state to launch comprehensive NaloxBox placement with 192 boxes including 51 in schools.
Maine reported 7,173 overdose reversals using state-supplied naloxone through its tiered distribution initiative.
West Virginia has seen over half of its harm reduction programs close due to restrictive regulations, with only 8 of 19 previously operational SSPs remaining. The The Cabell-Huntington Health Department (CHHD) in West Virginia is ended its 10-year-old syringe services program on December 16, 2025. Senate Bill 36 was introduced on January 14, 2026, with a goal of making syringe exchange programs unlawful.
Territorial Status:
Puerto Rico and U.S. Virgin Islands maintain syringe services programs.
No territories have authorized safe consumption sites.
Limited data available on territorial naloxone access and Good Samaritan law implementation.
7. LATEST NEWS
Russia Highlights Urgency of HIV Testing - According to the World Health Organization (WHO), Russia’s HIV prevalence is approximately 890 cases per 100,000 people. Their rate is higher than that of European countries and comparable to African countries such as Guinea and Liberia, which rank at 874 and 944, respectively. There is also concern that not all people living with HIV in Russia are in care; thus, there is a lapse in data between recorded cases and those in treatment. Transmissions continue to rise despite a drastic increase in testing since 2014. Health Minister Mikhail Murashko recommends increasing testing to one-third of the Russian population annually to curb the spread of the virus.
PEPFAR Data Indicate HIV Testing and Treatment Decline - Despite previous concerns that it would be completely dismantled, PEPFAR still exists. However, the harm caused by the current administration's previous freeze on funding are becoming apparent. Systemic interruption in treatment and care occurred when monies were held, and service providers were ordered to stop treatment with HIV medications obtained through PEPFAR, even if drugs had already been stocked. Treatment under PEPFAR remained about the same in the last quarter of 2025 as in the same span of time in 2024. Unfortunately, testing and new diagnoses have decreased along with the treatment of infants with HIV. The long-term effects of system disruptions caused by the arbitrary and capricious start and restart of the program are what have many experts worried.
NASTAD Publishes ADAP ‘State of the Union” - In February, NASTAD (National State Alliance of State and Territorial AIDS Directors) published its ADAP Watch, which tracks the current and projected fiscal health of ADAPs. The results come from 44 states and territories responding to a January 2026 RFI. Federal funding remains stagnant even though client enrollment and healthcare costs are steadily rising. Ten respondents reported budget deficits in the current fiscal year, while 19 ADAPs forecast deficits for the upcoming year. The main causes of budget shortfalls are increased drug costs per client, expiration of enhanced premium tax credits (leading to higher insurance costs), increased client enrollment, and decreased 340B rebates. Cost containment measures vary, including reductions in financial eligibility criteria, formulary reductions, reduced RWHAP Part B funding for core medical/support services, and monthly, annual, or per-client expenditure caps.
FDA Approves New Merck HIV Treatment - Recently, the FDA approved Merck’s new once-daily HIV single-pill regimen IDVYNSO (doravirine/islatravir). The treatment is important because it is the first and only non-INSTI, tenofovir-free, once-daily, complete two-drug regimen to demonstrate non-inferior efficacy in a head-to-head Phase 3 trial versus the three-drug regimen BIKTARVY® (BIC/FTC/TAF). It positions itself as a viable option for PLWHA who are virally suppressed but may need to switch from their current therapy. It also has a lowered risk for long-term toxicity, which is important given that the PLWHA are living longer. The clinical trials contained a large ethnic and racial variety of subjects with ages ranging from 18 to 80 years of age, with 11 percent being age 65 or older. As a complete regimen, no other HIV antiretrovirals are necessary. Although it is tolerated very well, there are a couple of drug interactions that are contraindicated as they could result in improper concentrations of islatravir or doravirine. IDVYNSO will be available in pharmacies after May 11, 2026.
Only 1 in 3 People Living With Hep C Are Being Treated - A recent research letter published in the Journal of the American Medical Association indicates that, despite the existence of curative direct-acting antiretrovirals (DAA), many people are not being treated. Only one-third receive treatment within one year of being diagnosed, 2.5 million to 4 million live with chronic infection, and the incidence of cases has increased during the past ten years. Researchers from the University of Virginia and Brigham and Women’s Hospital analyzed U.S. prescription data covering the time period January 2013 to December 2025. Since 2020, the treatment volume has been near the level of new U.S. infections. However, the number of treatment courses was significantly lower than what is needed to reach national elimination targets. Solutions include efforts to facilitate same-day treatment as well as telehealth and mobile outreach.
Florida ADAP Update - The initial cuts to the Florida ADAP program, which took effect on March 1, were slated to cause thousands of PLWHA to lose access to their HIV medication. March 24, 2026, the governor signed a bill to temporarily fund the ADAP program to support services through June 30, 2026. The rule that changed eligibility to 130% of the federal poverty level was reversed, changing it 400% (under $64,000 for an individual). Biktarvy is only available to those who are self-insured. Those who are uninsured can no longer receive it for direct dispensing. Additionally, Descovy is still covered, but as of April 1, 2026, it will require prior authorization for individuals with renal failure with a Creatinine Clearance (CrCL) less than 60. Notably, premium assistance for insurance has not been restored, but copay and deductible assistance are still available. Presently, the additional funding is only through June 30, 2026. Thus, the program's long-term stability remains a question.
What You Should Know About New Covid Variant - People living with chronic health conditions are at risk of more severe illness as a result of COVID-19 infections. A new Covid variant, SARS-CoV-2 B.A.32, has been spreading across the U.S. and at least 23 other countries as of February. There is no alarm or indication that infection by this variant is more severe than others. However, the variant contains mutations that make it likely to evade existing immunity. Antibodies generated from previous vaccinations or infections are proving less effective at neutralizing BA.3.2 compared to other existing Omicron variant strains. Notably, while it shows the ability to evade current protection, studies show it may be less effective at infecting cells. Importantly, thus far, it does not seem to be dominating the existence of other strains. Currently, scientists are monitoring to see if it develops any changes worthy of increased threat assessments. The symptoms of infection are no different from those of other strains.
Study Indicates Positive Mobile Telehealth Hep C Outcomes For Rural Drug Users - People who inject drugs (PWID) have a higher risk of Hepatitis C infection due to unclean needles. People with substance abuse issues in rural areas are at higher risk of transmission due to a lack of resources for treatment and testing, stigmatization, and a lack of drug treatment options. A group of researchers sought to examine the efficacy of a mobile telemedicine program for rural PWID. They parked a mobile van in places where PWID frequented. The study was conducted in rural New Hampshire and Vermont. The subjects were split into two groups. One group received all of their HCV care, including telemedicine visits on the mobile van. The other group only received treatment referrals by van staff and care navigation to a local or regional physician. Both groups were given access to sterile syringe exchange services. The group receiving complete care on the mobile van had a higher rate of HCV treatment initiation and a much higher rate of viral clearance than the alternate group.